"Under chronic stress, these same naturally-occurring steroids damage the protective functions of normal skin and inhibit wound healing, but during shorter intervals of stress, they are beneficial for inflammatory disorders and acute injury in both mice and humans," said senior investigator Peter Elias, MD, a UCSF professor of dermatology based at the San Francisco VA Medical Center (SFVAMC).
"We believe that our findings explain why this otherwise harmful component of the stress response has been preserved during human evolution," he said.
The study was published online in the Journal of Investigative Dermatology on August 7, 2014, in advance of print publication in the journal.
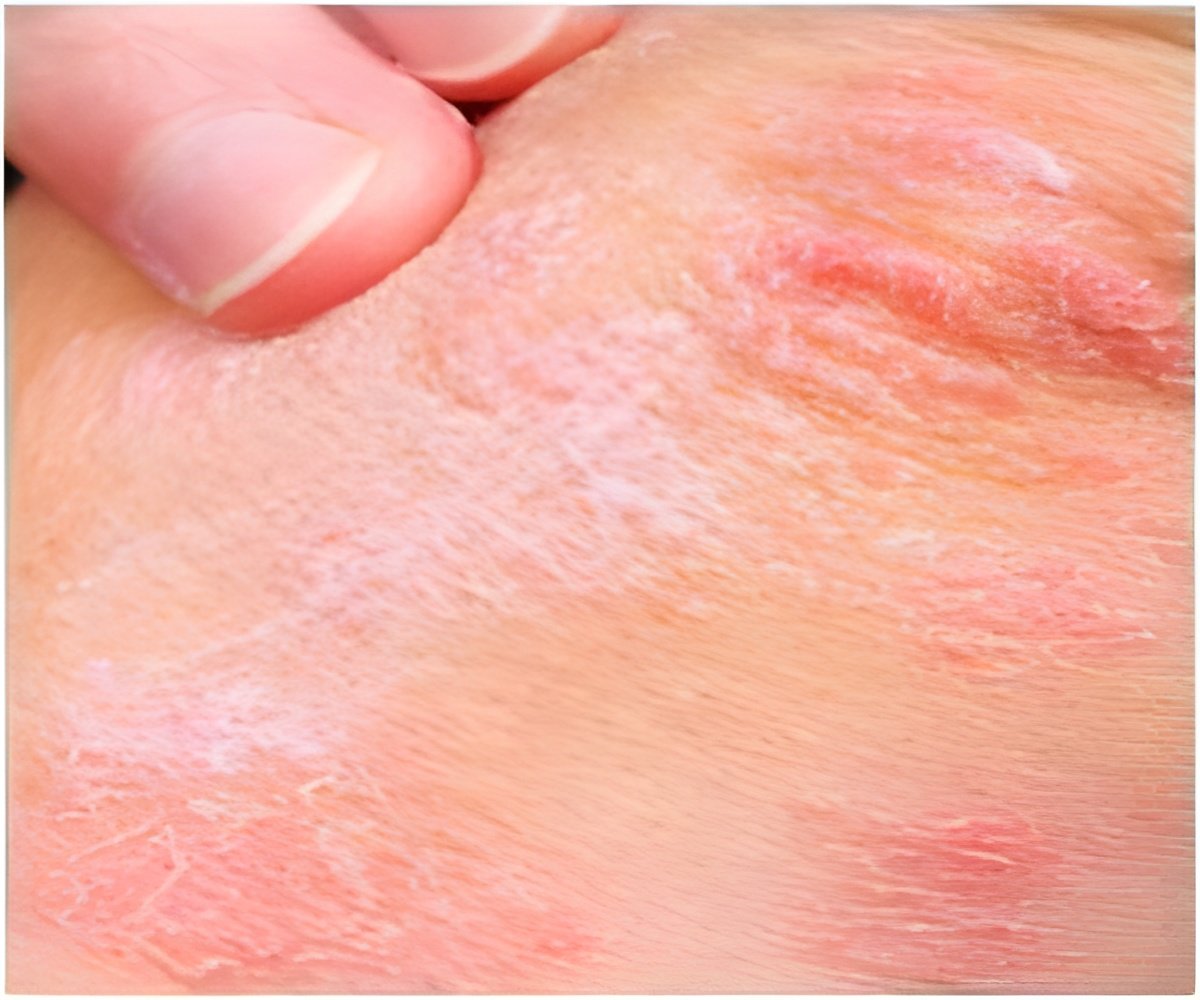
The scientists studied mouse models of three types of common skin irritations: irritant contact dermatitis, caused by exposure to an irritant such as a soap or solvent; acute allergic contact dermatitis, of the sort caused by poison ivy or poison oak; and atopic dermatitis, or eczema.
After exposure to irritants on a small patch of skin on one ear, one group of mice was returned to its regular cages, while another group was put in a stressful situation – being placed in very small enclosures for 18 hours a day over the course of four days.
Advertisement
When stressed mice were simultaneously given mifepristone, which blocks steroid action, all of the healing benefits of stress disappeared. "This demonstrated the central role of internal steroids in providing these benefits," said Elias.
Advertisement
According to Elias, the study provides a clue to an evolutionary puzzle: why, over millions of years, humans have preserved the tendency to produce steroids under stress. Previous research by Elias's laboratory and others has demonstrated that prolonged exposure to steroids harms both the structure and function of skin and other organs.
"Our ancestors did not have an arsenal of pharmaceutical steroids available to treat acute illnesses or injuries," Elias observed. "This safe, effective internal anti-inflammatory system provides just the correct amount of steroids to promote healing, over a time interval that is too short to cause harm."
Elias emphasized that the study did not look at the implications for human medical treatments. However, he contrasted the "substantial benefits" seen from modest increases in glucocorticoid levels brought on by short-term stress with the "adverse effects that we see all too commonly" with steroid therapy. Elias speculated that those negative effects could be the result of "overly aggressive treatment – too high doses, and perhaps for unnecessarily prolonged treatment intervals."
He said that while his research team did not study other kinds of inflammatory disorders, "the same benefits of psychological stress should accrue in any acute illness or injury."
Source-Eurekalert