‘Human brain’s tiniest blood vessels were found to trigger spinal motor neurons, prompting the neurons to grow during early development.’
Tweet it Now
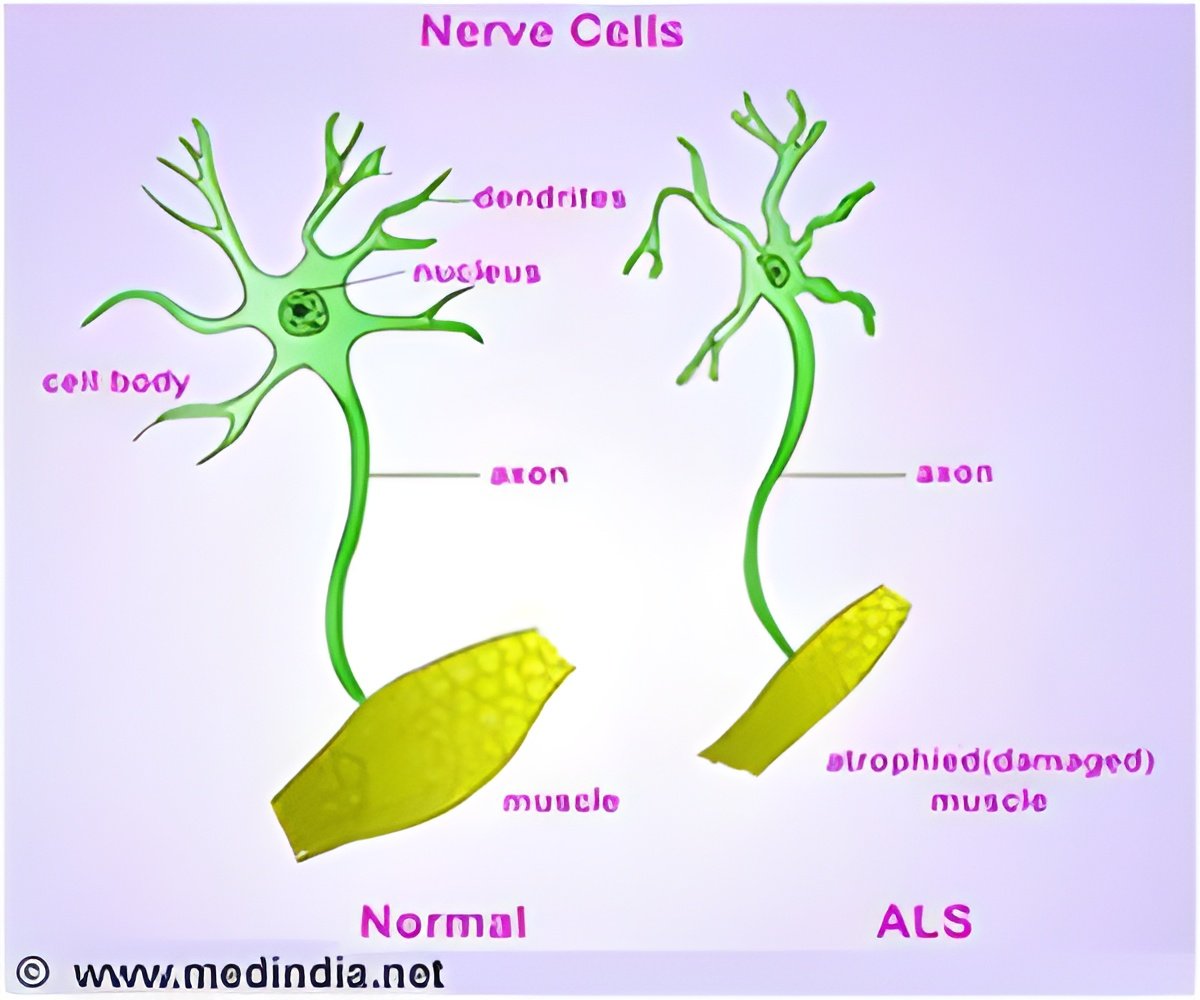
When a human embryo is about four weeks old, Sances explained, new blood vessels begin to surround a primitive column of cells that eventually will become the spinal cord. Driven by developmental genes, some of these cells turn into spinal motor neurons. The study showed the cells of the brain's smallest blood vessels, known as capillaries, are capable of activating these genes, which can spur spinal motor neurons to grow and mature. Besides providing insights into human biology, the study opened a new pathway to unraveling the mysteries of disorders such as ALS, or Lou Gehrig's disease, Sances said. ALS is a progressive, fatal disorder that kills motor neurons. There is no known cure. More than 6,000 people in the U.S. are diagnosed with the disorder each year, according to the ALS Association. "What may go wrong in the spinal neurons that causes the motor neurons to die?" Sances asked. "If we can model an individual ALS patient's tissues, we may be able to answer that question and one day rescue ALS patients' neurons through new therapies."
The study's findings were made possible by a unique pairing of stem cell science with Organs-on-Chips technology, which re-creates human biology in micro-engineered environments. Cedars-Sinai investigators first took samples of skin cells from adults and genetically reprogramed them into induced pluripotent stem cells, which can create any type of cell--in this case, spinal motor neurons and the lining of the brain's capillaries. The team placed these cells in the tiny channels of Organ-Chips, which are made of flexible polymer and are about the size of AA batteries. In the chips, nurtured by special fluids, the cells of the two different tissues thrived and interacted with each other.
"This study told us something important about how our neurons develop," said Clive Svendsen, PhD, professor of Medicine and Biomedical Sciences, director of the Cedars-Sinai Board of Governors Regenerative Medicine Institute and senior author of the study. As a next step, he added, investigators are developing plans to use chip technology to compare the vessel-neuron interactions in ALS patients against those of individuals without ALS. The research is part of the new Patient-on-a-Chip program, a collaboration between Cedars-Sinai and Emulate Inc. in Boston to help predict which disease treatments would be most effective based on a patient's genetic makeup and disease variant. Emulate produces the Organ-Chips used in the program. Geraldine A. Hamilton, PhD, Emulate's president and chief scientific officer, is a co-author of the spinal motor neuron study. In February, investigators from the two organizations announced they had used an Intestine-Chip to model a human intestinal lining.
The Patient-on-a-Chip program is an important initiative of Cedars-Sinai Precision Health, whose goal is to drive the development of the newest technology and best research, coupled with the finest clinical practice, to rapidly enable a new era of personalized health
Advertisement