‘Up to 60% of those with thyroid disease are unaware of their condition. Women are five to eight times more likely than men to have thyroid problems.
’
Tweet it Now
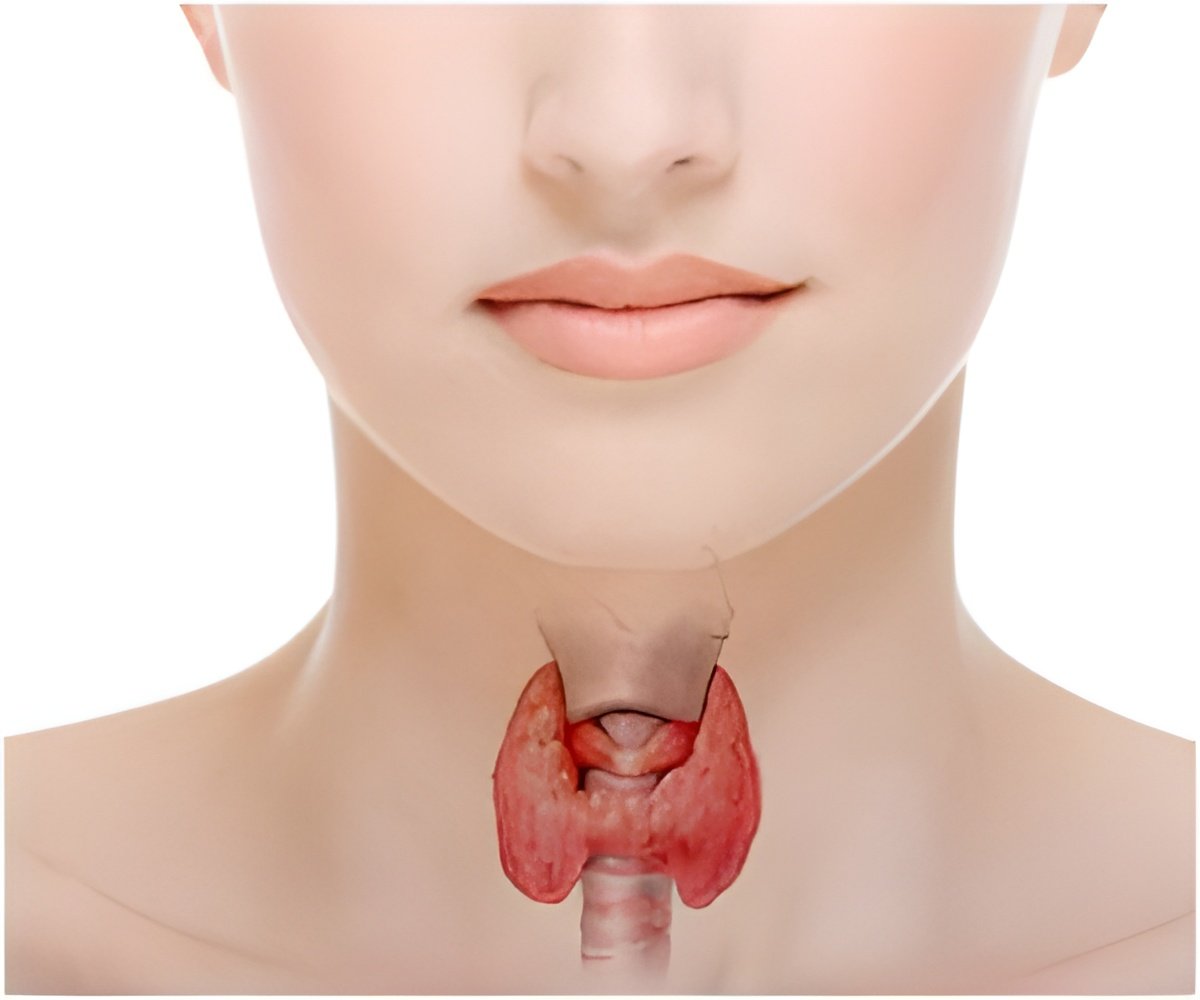
The thyroid gland is a hormone factory. Under normal conditions, the factory foreman -- the molecule calling the shots -- is the aptly named thyroid-stimulating hormone. Called TSH for short, this hormone attaches to a receptor on the thyroid cell surface, triggering a series of signals that provoke the gland to pump out thyroid hormones. However, sometimes autoantibodies, essentially posing as TSH, attach to the receptor and trick the thyroid into flooding the body with more hormones than are needed. In addition, sometimes the receptor itself has an inherited or acquired mutation that triggers production of too many or too few hormones.
Torsten Schöneberg of the University of Leipzig in Germany, who led the study reported in the JBC, says it was curious that the legitimate activator hormone, autoantibodies and mutations all were able to provoke thyroid hormone production. So his team set out to identify a common thread among them.
Turns Out, The Answer Lay In The Receptor Itself
"We discovered a small amino acid sequence - we call it p10 because it is 10 amino acids long -- within the TSH receptor protein," Schöneberg explains. This sequence "functions as activator of the receptor upon binding of the hormone or autoantibodies."
Advertisement
"In most other hormone-receptor systems, the hormone directly activates the receptor protein," says Antje Brüser, first author of the study and junior scientist of the group, offering the example of how adrenalin activates the ß adrenergic receptor. "In the case of glycoprotein hormone receptors, the family of proteins that includes the thyroid receptor, upon binding of the extracellular hormone, the intramolecular activator (p10) induces structural changes of the receptor protein, triggering activation of the intracellular signaling cascade."
Advertisement
Source-Eurekalert