"It's well known that, in mammals, these specialized sensory cells don't regenerate after damage," said Alan Cheng, MD, assistant professor of otolaryngology. (In contrast, birds and fish are much better equipped: They can regain their sensory cells after trauma caused by noise or certain drugs.) "Identifying the progenitor cells, and the cues that trigger them to become sensory cells, will allow us to better understand not just how the inner ear develops, but also how to devise new ways to treat hearing loss and deafness."
The research will be published online Feb. 26 in Development. Cheng is the senior author. Former medical student Taha Jan, MD, and postdoctoral scholar Renjie Chai, PhD, share lead authorship of the study. Roel Nusse, PhD, a professor of developmental biology, is a co-senior author of the research.
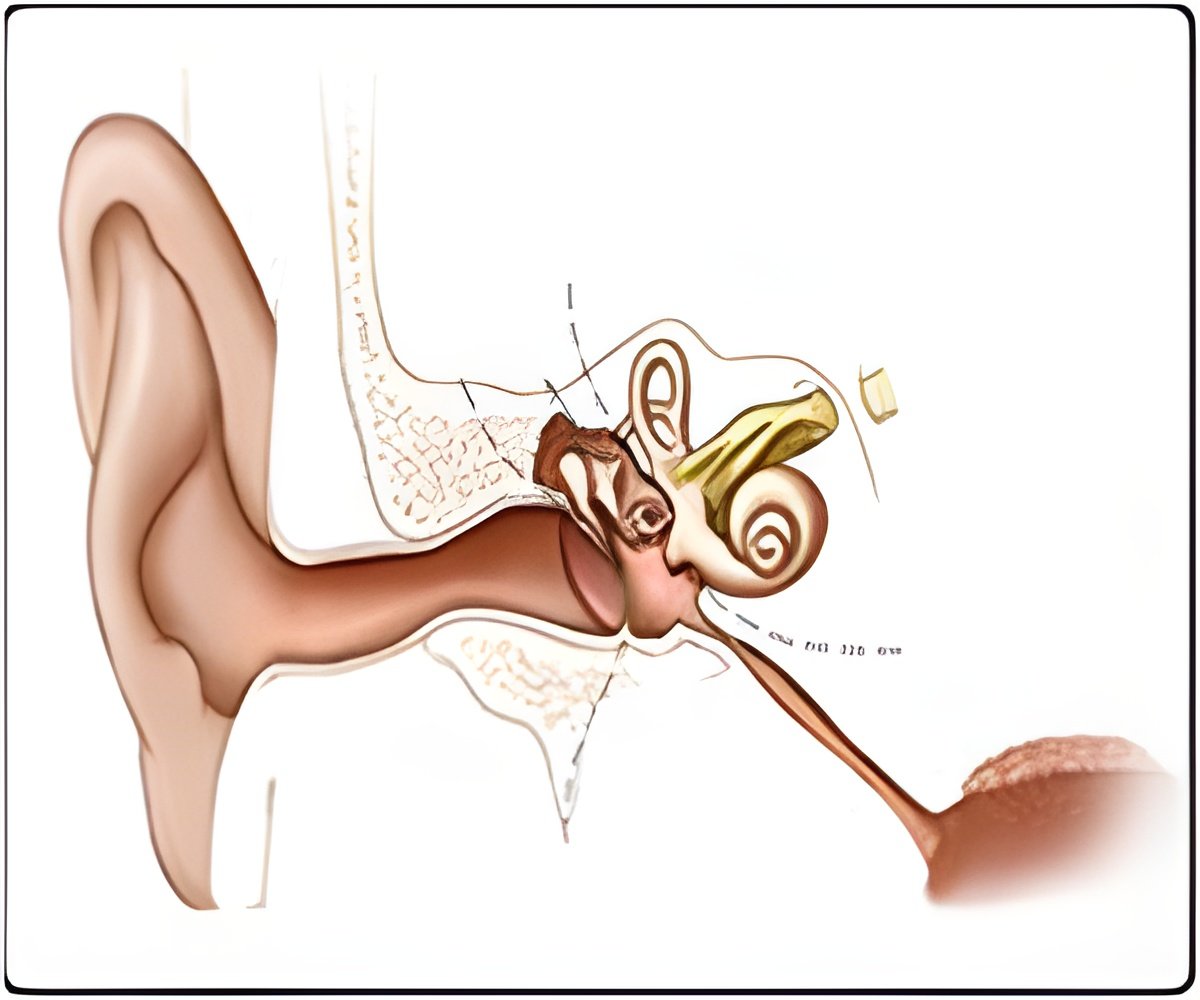
The inner ear is a highly specialized structure for gathering and transmitting vibrations in the air. The auditory compartment, called the cochlea, is a snail-shaped cavity that houses specialized cells with hair-like projections that sense vibration, much like seaweed waving in the ocean current. These hair cells are responsible for both hearing and balance, and are surrounded by supporting cells that are also critical for hearing.
Twenty percent of all Americans, and up to 33 percent of those ages 65-74, suffer from hearing loss. Hearing aids and, in severe cases, cochlear implants can be helpful for many people, but neither address the underlying cause: the loss of hair cells in the inner ear. Cheng and his colleagues identified a class of cells called tympanic border cells that can give rise to hair cells and the cells that support them during a phase of cochlear maturation right after birth.
"Until now, these cells have had no clear function," said Cheng. "We used several techniques to define their behavior in cell culture dishes, as well as in mice. I hope these findings will lead to new areas of research to better understand how our ears develop and perhaps new ways to stimulate the regeneration of sensory cells in the cochlea."
Advertisement
Although regeneration of sensory hair cells does not happen naturally, recent research has suggested that the mammalian ear may harbor a sub-population of — presumably inactive — progenitor cells. The research team led by Cheng and Nusse used a strain of laboratory mice that allowed the scientists to track the activation of a cell-signaling pathway driven by a protein called Wnt. The Wnt pathway has previously been shown to be involved in many developmental functions, and it drives the renewal and proliferation of many types of stem cells.
Advertisement
The researchers found that tympanic border cells, or TBCs, which form a thin layer under the sensory epithelium, are actively dividing in mice during the first three weeks after birth (the time corresponding to about the first trimester of human development, during which the ability to hear is established) and give rise to at least a subset of sensory and non-sensory cells in the ear. They also divided vigorously in isolated cochlea when the Wnt pathway was activated, and stopped when the pathway was inhibited. Finally, the researchers showed that purified TBCs were able to specialize into hair cells and support cells when cultured in a laboratory dish.
"It's surprising to think that these progenitor cells are among this largely underappreciated group of cells," said Cheng. "This study also highlights that, even in mice, there is a lot of maturation occurring after birth as hearing develops. There's clearly a lot more to be understood. Next we'd like to look at these cells in models of hearing loss. Do they have the ability to regenerate? If so, under what conditions?"
Source-Eurekalert