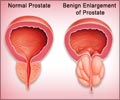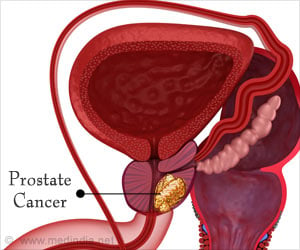
In order to determine if a more frequent screen detection in PIVOT explains the lower AMD when compared to the SPCG-4 trial, Jing Xia, Ph.D., of the Fred Hutchinson Cancer Research Center in Seattle and colleagues, assumed that the SPCG-4 trial represented RP efficacy and prostate cancer survival in an unscreened population. They then adjusted prostate cancer survival using published estimates of overdiagnosis and lead time to evaluate the effect of screen detection on disease-specific deaths and the observed AMD.
The researchers found that overdiagnosis and lead time explains the lower AMD in PIVOT if the RP efficacy and prostate cancer survival in the absence of screening are comparable to that of the SPCG-4 trial. They conclude that if these findings are the correct explanation, then a specific set of cases should not be treated with RP and that their identification should lead to a better understanding of the RP benefit in the remaining cases. "PIVOT should not be interpreted as evidence that RP is not efficacious in reducing prostate cancer mortality," the researchers write. "PIVOT should encourage us to develop tests to identify cases for which immediate treatment is beneficial."
Source-Eurekalert



![Prostate Specific Antigen [PSA] & Prostate Cancer Diagnosis Prostate Specific Antigen [PSA] & Prostate Cancer Diagnosis](https://www.medindia.net/images/common/patientinfo/120_100/prostate-specific-antigen.jpg)