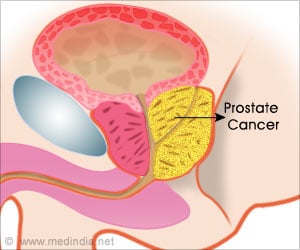
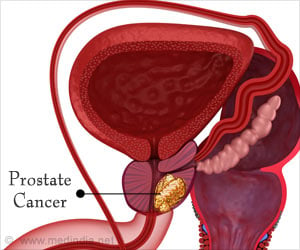
‘IsoPSATM test can identify molecular changes in prostate specific antigen (PSA) protein more precisely and also differentiate between high-risk and low-risk disease.
’
Tweet it Now
Although widely used, the current PSA test relies on detection strategies that have poor specificity for cancer - just 25 percent of men who have a prostate biopsy due to an elevated PSA level actually have prostate cancer, according to the National Cancer Institute - and an inability to determine the aggressiveness of the disease. The IsoPSA test, however, identifies prostate cancer in a new way. Developed by Cleveland Clinic, in collaboration with Cleveland Diagnostics, Inc., IsoPSA identifies the molecular structural changes in protein biomarkers. It is able to detect cancer by identifying these structural changes, as opposed to current tests that simply measure the protein's concentration in a patient's blood.
"While the PSA test has undoubtedly been one of the most successful biomarkers in history, its limitations are well known. Even currently available prostate cancer diagnostic tests rely on biomarkers that can be affected by physiological factors unrelated to cancer," said Eric Klein, chair of Cleveland Clinic's Glickman Urological & Kidney Institute. "These study results show that using structural changes in PSA protein to detect cancer is more effective and can help prevent unneeded biopsies in low-risk patients."
The clinical trial involves six healthcare institutions and 132 patients, to date. It examined the ability of IsoPSA to distinguish patients with and without biopsy-confirmed evidence of cancer. It also evaluated the test's precision in differentiating patients with high-grade (Gleason =7) cancer from those with low-grade (Gleason = 6) disease and benign findings after standard ultrasound-guided biopsy of the prostate.
Substituting the IsoPSA structure-based composite index for the standard PSA resulted in improvement in diagnostic accuracy. Compared with serum PSA testing, IsoPSA performed better in both sensitivity and specificity.
Advertisement
"In general, the clinical utility of prostate cancer early detection and screening tests is often limited by the fact that biomarker concentrations may be affected by physiological processes unrelated to cancer, such as inflammation, as well as the relative lack of specificity of these biomarkers to the cancer phenotype. In contrast, clinical research data suggests that the IsoPSA assay can interrogate the entire PSA isoform distribution as a single stand-alone diagnostic tool which can reliably identify structural changes in the PSA protein that correlate with the presence or absence and aggressiveness of prostate cancer."
Advertisement