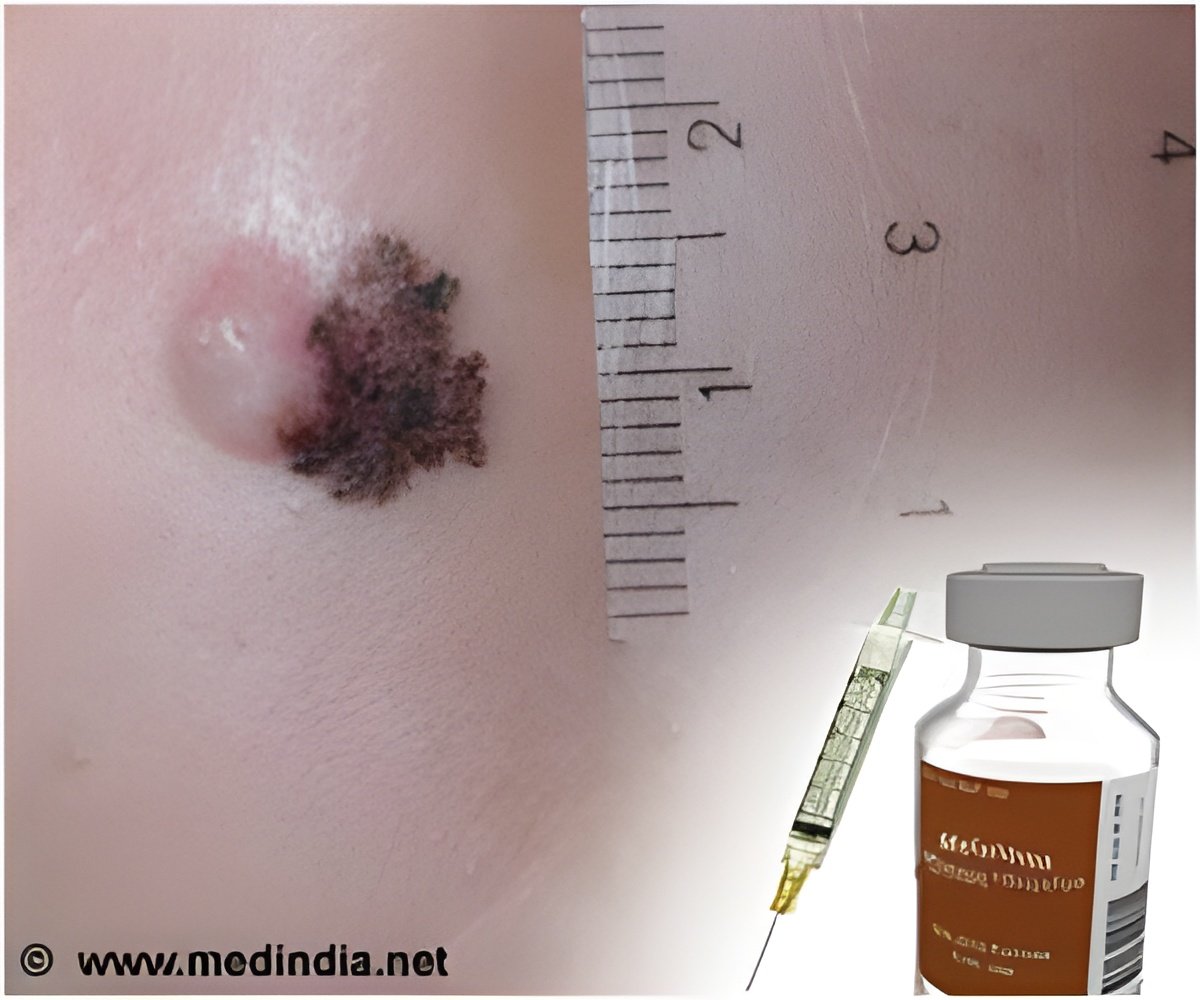
The study involved 951 patients with stage III melanoma, many of whom faced a likely recurrence of cancer since it had spread to their lymph nodes.
Three years after treatment, the survival rate was 46.5 percent in the ipilimumab group and 34.8 percent in the placebo group.
Ipilimumab has been approved by the US Food and Drug Administration for the treatment of inoperable stage IV melanoma. It works by disarming the tumor's defense against the immune system.
"This is a promising treatment -- we saw substantially fewer recurrences among patients who are at high risk of relapse," said lead study author Alexander Eggermont, director general of the Gustave Roussy Cancer Center in Paris, France.
"This trial with ipilimumab is the first to show we may be able to give these drugs earlier in the course of disease, where they can do more good and potentially cure more patients."
Advertisement
However, researchers cautioned that side effects were "considerable," and included five people who died and were classed as "treatment-related deaths," according to a summary of the study provided to the media.
Advertisement
Another clinical trial, this one an early phase I trial involving 411 patients with advanced melanoma, showed promising results using a Merck-made therapy called a PD-1 targeting antibody MK-3475.
The Food and Drug Administration has granted breakthrough therapy designation for the agent, which stimulates the immune system in a different way than Yervoy.
A total of 34 percent of patients in the study showed some response to the treatment, meaning that their tumors grew smaller.
In all, 69 percent of patients were alive after one year.
"These are early data, but they tell us something really important," said lead study author Antoni Ribas, a professor of medicine at the University of California, Los Angeles.
Eight percent of patients experienced serious side effects related to the treatment, but just four percent dropped out of the study as a result.
A third study on 94 patients with inoperable melanoma showed that a combination of ipilimumab and nivolumab, both made by Bristol Myers-Squibb, allowed a midpoint survival of three and a half years, or double what previous studies using each drug alone had shown.
"Just a few years ago, median survival for patients diagnosed with advanced melanoma was as little as a year or less, and only approximately 20-25 percent survived two years," said lead study author Mario Sznol, professor of medical oncology at Yale University.
"So it's truly remarkable that we're seeing a median survival over three years in this trial," he said.
Long term data was available for 53 patients, of whom 22 (41 percent) responded to the treatment and nine (17 percent) had complete remissions.
"While we are encouraged by what we are seeing with these two drugs together, this trial was small, so a randomized phase III trial will be important to validate our initial results," said Sznol.
Source-AFP












