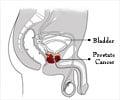
The results being presented are a culmination of three clinical studies. The first study identified 12 biomarkers that predicted both lethal outcome and prostate cancer pathology, and of those, eight were targeted in a clinical development biopsy study as the final subset for the ProMark™ test.
The third, blinded clinical validation study of ProMark™, which analyzed 274 prostate cancer biopsies, accurately predicted the overall prostate cancer pathology for patients with biopsy Gleason grades of 3+3 (=6) or 3+4 (=7). Quantitative measurements on the biopsy samples allowed researchers to successfully identify ProMark™ risk scores for 'favorable' cases (surgical Gleason score 3+3 or 3+4; organ-confined disease [<=pT2]) as potential candidates for active surveillance and 'unfavorable' cases (≥T3a, N, or M or surgical Gleason >=4+3), more likely in need of aggressive therapy, with an AUC of 0.69 (0.61-0.74; p<0.0001) and odd's ratios lowest to highest quartile of 5.
Researchers found that patients with low-risk ProMark™ scores (<0.33) were accurately identified as having favorable disease 81 percent of the time, with 90 percent specificity, while high-risk scores (>0.8) indicating non-favorable disease were predicted 77 percent of the time. Importantly, the ProMark™ test was found to provide additional, independent prediction relative to standard risk-stratification systems, including National Comprehensive Cancer Network (NCCN) guidelines, the D'Amico system, and CAPRA, all using Prostate-Specific Antigen (PSA) levels, Gleason grades and T stage to group men as low, intermediate, or high-risk.
"A key challenge in treating men with prostate cancer is determining whether they need aggressive therapy, or if active surveillance is appropriate. Current clinical and pathological parameters are insufficient in predicting the risk of aggressive cancer for early stage cancer patients" said Peter Blume-Jensen, M.D., Ph.D., Senior Vice President and Chief Scientific Officer at Metamark. "ProMark™ was developed with the specific purpose to address this need. This latest data is a significant milestone for Metamark as it indicates ProMark™ can be used as an objective aid in decision making together with standard-of-care for prostate cancer prognosis, and thereby help maintain a higher quality of life for patients by avoiding unnecessary aggressive therapy, like surgical prostatectomy or radiation."
Advertisement



![Prostate Specific Antigen [PSA] Prostate Specific Antigen [PSA]](https://www.medindia.net/images/common/patientinfo/120_100/prostate-specific-antigen.jpg)