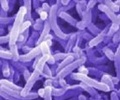
ETEC is also the leading cause of illness among international travelers to developing countries. Among the 65 million people travelling from industrialized countries to ETEC high-risk areas in the developing world, 30-50 % (ca 30 million people) experience at least one TD episode during their journey.
Today there is no ETEC vaccine available on the market for use in either children or travelers in ETEC high-risk areas.
A novel oral ETEC vaccine has been developed in Sweden and a clinical phase I-study of this vaccine has now been successfully completed. All predefined primary endpoints for the study were achieved showing that the vaccine is safe, with only few and mild adverse reactions observed, and strongly immunogenic, stimulating immune responses to all vaccine components.
The vaccine, consisting of four inactivated E. coli bacterial strains and the ETEC-based B subunit protein LCTBA, was studied for safety and immunogenicity in a total of 129 healthy volunteers. The study was designed as a four-armed, double-blinded, placebo-controlled trial. Study participants were randomized to receive two oral doses two weeks apart of either the vaccine alone, vaccine co-administered with an oral adjuvant in a low or a higher dose, or an inactive placebo preparation (a buffer solution).
In addition to safety parameters, antibody responses against various vaccine components in fecal samples, or antibodies produced by intestine-derived peripheral blood lymphocytes were measured at different time intervals.
Advertisement
Furthermore, 85 % of subjects receiving the vaccine co-administered with low dose adjuvant showed significant immune responses to all vaccine components. In addition to showing excellent safety and immunogenicity of the five-component vaccine, the results also confirm previous preclinical results as well as clinical results obtained with a prototype version of this vaccine.
Advertisement
Source-Eurekalert