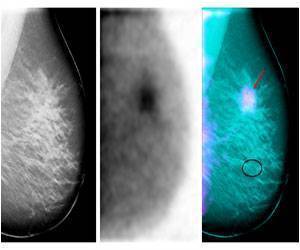
Talazoparib, a PARP inhibitor could be effective in treating against certain triple-negative breast cancers (TNBC) that lack BRCA mutation.

‘Three PARP inhibitors have been approved so far by the U.S. Food and Drug Administration to treat certain Triple-Negative Breast Cancers (TNBC).’





The authors are Funda Meric-Bernstam, MD, chair of the Department of Investigational Cancer Therapeutics, medical director of the Institute for Personalized Cancer Therapy, and Nellie B. Connally Chair in Breast Cancer Research at The University of Texas MD Anderson Cancer Center in Houston."Patients with triple-negative breast cancer who do not respond to neoadjuvant [preoperative] chemotherapy have a decreased chance of long-term survival, and there is a great need for new treatment options," said Meric-Bernstam.
"Developing animal models that are well characterized gives us an opportunity to test new agents and better understand how to best treat these patients."
Because PDXs are tumor models derived directly from patient tumors and grown in mice, they may better reflect the complexity of human tumors than traditional models, she explained.
"Ultimately, we would like to better deliver personalized cancer therapy by understanding how we can best match therapies to patients," she said.
Advertisement
"The PDX models we generated varied dramatically in their genomic characteristics, potentially giving insight into why it has been challenging to truly personalize cancer therapy," Meric-Bernstam noted.
Advertisement
"Among targeted therapies, the efficacy of PARP inhibitors was most striking," noted Meric-Bernstam. Of the PDXs treated with talazoparib, five regressed and one showed stable disease for at least 28 days. Four of these five PDXs did not harbor germline BRCA1/2 mutations, but had alterations in other DNA repair pathways, including ATM deletion and other alterations in BRCA2.
The team is developing new PDX models of TNBC to better determine which models are sensitive to PARP inhibitors and to get a more comprehensive picture of the determinants of PARP inhibitor sensitivity. They are also conducting an investigator-initiated clinical trial to test whether talazoparib is efficacious in other tumor types with DNA damage repair alterations beyond germline BRCA mutations.
The U.S. Food and Drug Administration has approved three PARP inhibitors so far, olaparib (Lynparza) and rucaparib (Rubraca) to treat certain women who have ovarian cancer with BRCA gene mutations, and niraparib (Zejula) to treat both BRCA mutation-positive and BRCA mutation-negative ovarian cancers.
"Our studies show that PARP inhibitors may have efficacy in selected patients outside of germline BRCA mutation, potentially those with other alterations in DNA damage repair genes," Meric-Bernstam said.
A limitation of the study is that the researchers used a limited number of PDX models and the findings need to be validated in a larger panel of PDXs, Meric-Bernstam said.
Source-Eurekalert