‘Paclitaxel-coated and paclitaxel-eluting stents and balloons devices are safe in patients with lower extremity artery disease (LEAD).
’
Tweet it Now
However, the current study has analyzed data on the use of paclitaxel-coated stents and balloons in 64,771 'real world' patients with lower extremity artery disease (LEAD) who underwent 107,112 procedures to unblock arteries from the time that paclitaxel was first used to coat stents and balloons in 2007 up to 2015. The patients continued to be followed until 2017 - a follow-up period of more than 11 years, which is longer than any other study.The first author of the EHJ paper, Dr. Eva Freisinger, of the University Hospital Münster, Germany, said: "Our findings show that paclitaxel-based devices are safe and are not associated with an increase of death. To our knowledge, this is the largest real-life group of patients that have been evaluated with long-term follow-up from the time that paclitaxel-coated devices were first introduced. Our work provides a solid base of evidence that will be difficult to rebut. We expect the FDA and other regulatory authorities will very likely amend their statements on safety concerns on paclitaxel-based devices."
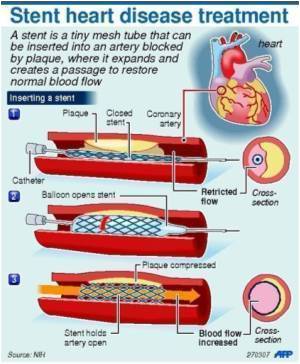
Paclitaxel is an agent that prevents the proliferation of cells; it is used for coating stents (tubes) and balloons because it limits the growth of scar tissue and a recurrence of blockages in the treated arteries (restenosis). Drug-eluting stents and balloons have been used in millions of patients with lower extremity artery disease worldwide, with over 55,000 balloons and 6,600 drug-eluting stents implanted annually in Germany alone, of which 97% use paclitaxel.
The concerns raised by the meta-analysis at the end of 2018 and the subsequent FDA letter of concern to healthcare providers, resulted in an estimated 50% drop in their use
In the current study, researchers in Germany retrieved data from 9.2 million people in the German health insurance scheme, BARMER, to identify all patients who had first endovascular revascularisation for lower extremity artery disease between 2007 and 2015. The patients were treated with one or more of the following:
Advertisement
- paclitaxel drug-eluting stents;
- paclitaxel-coated balloons;
- bare-metal stents (i.e., stents that were not coated with a drug);
- plain old balloon angioplasty' (i.e., balloons that were not coated with a drug).
None of the patients had been treated previously with paclitaxel-coated devices because these were only introduced in 2007. However, during the follow-up period to 2017, many of the patients may have had more than one type of device fitted in subsequent procedures, and the researchers took this into account in their analyses. During this time, a total of 23,137 drug-eluting devices were used. By 2017 42% of patients had died.
After adjusting their analyses to take account of factors that could affect the results such as age, sex, pre-existing heart or blood vessel risk factors (such as smoking and weight) and other medical conditions, the researchers found that the risk of death from any cause up to 30 days after the revascularisation did not differ significantly between the different devices used.
Advertisement
The authors write: "Based on 9.2 million insurants, respectively over 10% of the German population, our data represent the current practice in endovascular treatment of LEAD."
Dr. Freisinger said: "Our study illustrates the value of research using data from health services to assess safety concerns in real-life groups of patients rapidly."
The strength of the study is the very large number of patients, with full and comprehensive data on them all and long follow-up. Limitations include the fact that the data do not give information on the underlying reason for treatment with a drug-eluting device.
Source-Eurekalert