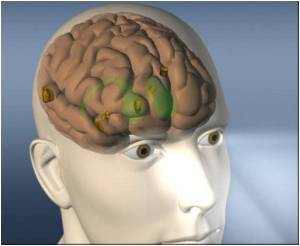
NovoTTF system for glioblastoma tumors is capable of giving electric currents to disrupt tumor cells before they divide. FDA is yet to give the final approval for its use on patients with glioblastoma tumors.
U.S. Food and Drug Administration (FDA) Neurological Devices Advisory Panel is of the opinion that the risks of using the NovoTTF-100A System (NovoTTF) is reduced when it is administered as monotherapy instead of standard medical therapy.
A randomized phase III trial for 237 patients with glioblastoma tumors showed that patients treated with the NovoTTF were benefited with better survival time than patients treated with chemotherapy. NovoTTF treated patients also said that they had better quality of life and lesser side effects compared to those treated with chemo.
Source-Medindia