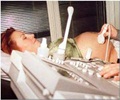
Patients suffering from major depression now have a safe, effective, non-drug treatment, thanks to Rush University Medical Center’s Transcranial Magnetic Stimulation (TMS) Clinic.
TMS therapy is the first FDA-approved, non-invasive antidepressant device-based treatment clinically proven for treatment of depression.Psychiatrists led by Dr. Philip Janicak at Rush were among the first to test the technique.
The TMS therapy system delivers highly focused magnetic field pulses to a specific portion of the brain, the left prefrontal cortex, in order to stimulate the areas of the brain linked to depression.
The repeated short bursts of magnetic energy introduced through the scalp excite neurons in the brain.
Janicak said that drug treatment options could be ineffective or intolerable due to side effects.
Current antidepressant therapies are not beneficial for at least a third of depressed individuals, leaving many with a lack of adequate treatment options.
Advertisement
TMS therapy does not require anesthesia or sedation and patients remain awake and alert.
Advertisement
"TMS therapy is a safe and effective alternative for patients who suffer from major depressive disorder and are not getting satisfactory improvement from antidepressant medications," said Janicak.
Source-ANI
ARU