Novartis had narrowed down the benefits in its approval application before successfully presenting it to the FDA and is now looking forward towards a similar result in Europe after having filed applications with the European Medicines Agency and the Swiss regulator.
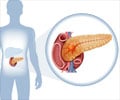
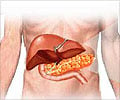
Stating that their drug was the first to be approved for the disease for more than three decades in the United States, Novartis released a statement saying, “Data show Afinitor delays tumor growth and reduces risk of disease progression in patients with advanced neuroendocrine tumors (NET) of pancreatic origin.”
Source-Medindia