‘The Medical Director of the GTB Hospital has stated that the samples of the injection have been sent to the microbiology lab for testing so that suspected contamination could be determined.’
Tweet it Now
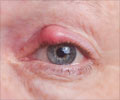
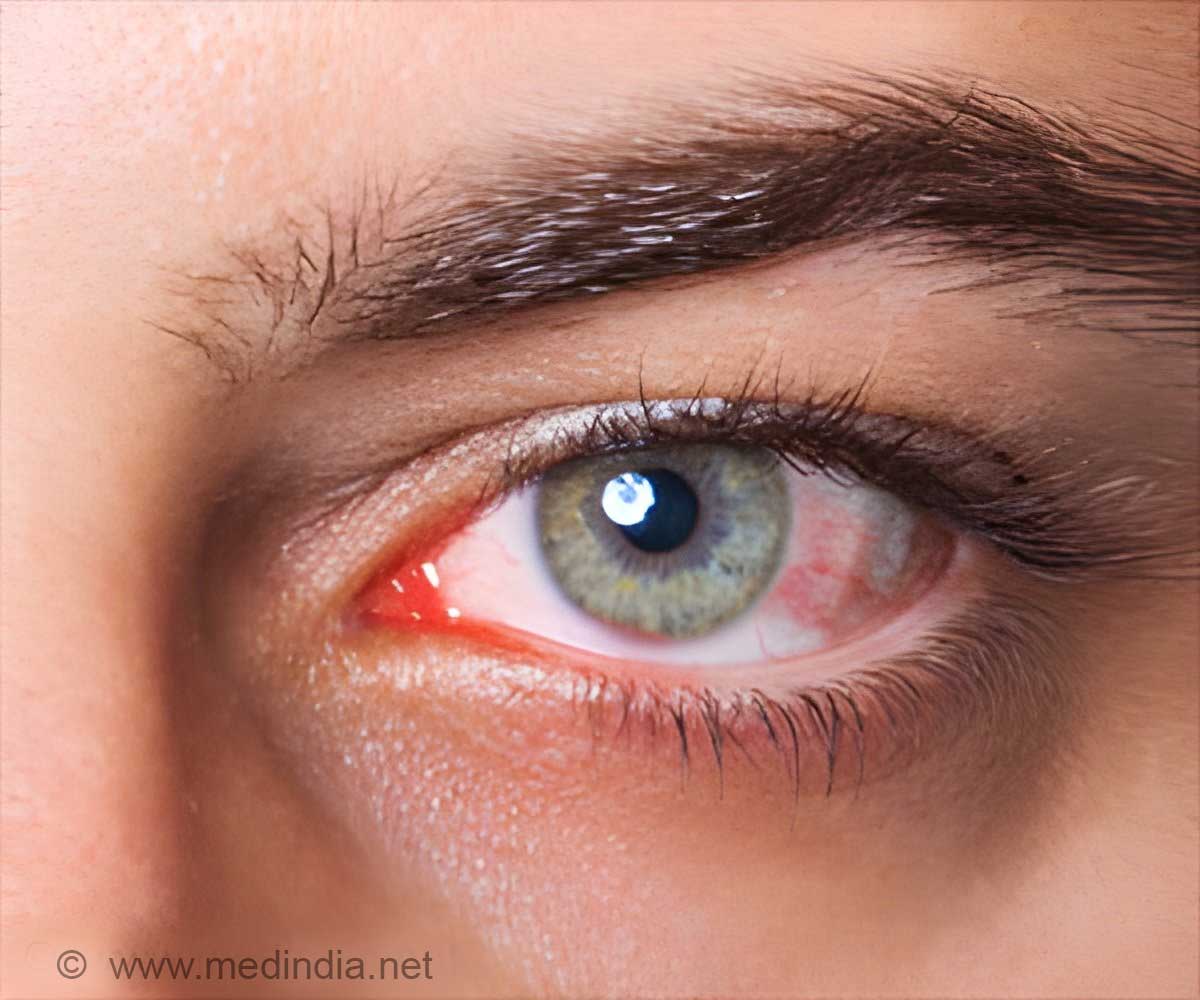
About eight of them reportedly require eye surgery. "The commission has observed that the contents of the news report raise serious issue of negligence by the GTB Hospital doctors and its management, putting the eyesight of the patients to risk."
"Accordingly, it has issued a notice to the Chief Secretary, Delhi government, calling for a detailed report in the matter within four weeks," the statement said.
The statement said patients, as part of their follow-up treatment, were administered injections containing the drug "Avastian", which is globally used to prevent blindness due to ageing, diabetes, hypertension and rupturing of blood vessels.
"Reportedly, Atul Kumar, Chief of the R.P. Centre for Ophthalmic Sciences at AIIMS, has said that 'Avastian' is classified as sight saving drug and used all over the world. In this case, it seems that the injections were contaminated, which caused the reaction," it said.
Advertisement
"He has informed that specialists were called immediately when the patients complained about blurred vision and mild pain, during the follow up and the serious cases were referred to the AIIMS," the statement added.
Advertisement