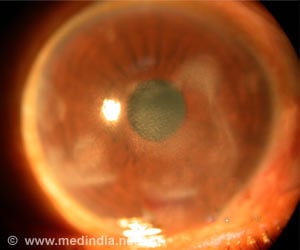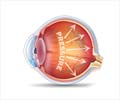
The article "Intraocular Pressure Homeostasis: Maintaining Balance in a High-Pressure Environment," by Ted Acott and coauthors, Oregon Health & Science University, Portland, OR, describes the efficient mechanisms at work in the eye to keep intraocular pressure within an acceptable range for 92-98% of the population. Understanding these mechanisms will enable the development of drug interventions to treat the unfortunate 2-8% of people that are at risk of developing elevated eye pressure and glaucoma.
"The TM, a unique multilayered tissue that controls intraocular pressure, and its surrounding structures represent viable targets for the development of novel glaucoma therapies," write Editor-in-Chief W. Daniel Stamer, PhD, Duke University (Durham, NC) and Guest Editor John R. Samples, MD, Professor, Rocky Vista University and Director, Western Glaucoma Foundation, Portland, OR, in the Editorial "The Trabecular Meshwork Special Issue, Inspired by the TM Study Club."
The special double issue provides a comprehensive look at the TM and next-generation glaucoma therapies in development through a collection of editorials, original research articles, and reviews. Included is the review article "The Role of TGF-ß2 and Bone Morphogenetic Proteins in the Trabecular Meshwork and Glaucoma," in which Robert Wordinger, Tasneem Sharma, and Abbot Clark, University of North Texas Health Science Center, Fort Worth, describe the TGF-ß superfamily of growth factors and their role in primary open-angle glaucoma (POAG), the second leading cause of blindness worldwide.
Also of note, Nelson Winkler, Mayo Clinic College of Medicine, and Michael Fautsch, Mayo Clinic, Rochester, MN, explore the current understanding of how prostaglandin analogues, first-line treatments for glaucoma, work to reduce elevated intraocular pressure, in the review article "Effects of Prostaglandin Analogues on Aqueous Humor Outflow Pathways."
Source-Eurekalert