Mayo Clinic in Florida expects to offer the new treatment immediately, says C. Daniel Smith, M.D., chair of the Surgery Department at Mayo Clinic in Florida, and an internationally recognized expert on the treatment of GERD.
Dr. Smith is experienced in using the system because Mayo Clinic in Florida was one of only 14 centers nationally that participated in a clinical trial that led to the FDA’s approval of the device.
“Mayo has been a leader in the treatment of esophageal diseases, especially GERD, and we are pleased to be offering this new treatment to our patients immediately,” he says.
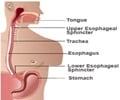
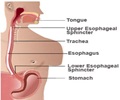
GERD is a condition in which liquid, or food, in the stomach flows back up into the esophagus due to the inability of a ring of muscle between the lower esophagus and the top of the stomach to close properly.
If drugs aimed at neutralizing the acid in the stomach fails to prevent GERD, an operation designed to correct the mechanical defect is considered. But between 1.5 million and 2 million patients of those patients could benefit from treatment that is much less complex than current surgical options, Dr. Smith says.
Advertisement
The results of the clinical study that led to approval of the device have not yet been published. But “the data presented to the FDA revealed striking results when compared to other GERD treatments that have been investigated over the past 20 years,” Dr. Smith says. “The system offers effective control of GERD with limited side effects and thus far an excellent safety record.”
Advertisement
Dr. Smith performs about 200 GERD-related surgeries a year and has been involved with many new treatments over the past several decades. “I expect this device to be a game changer for the treatment of GERD in select patients who have failed management with drugs,” says Dr. Smith.
Ken DeVault, M.D., chair of the Department of Internal Medicine at Mayo Clinic in Florida, also participated in these studies. “I have many patients who are searching for something more than medication for their reflux, but have been hesitant to undergo a traditional reflux surgery,” he says. “I think this procedure may well be a very attractive option for that group.”
Mayo Clinic physicians and scientists collaborated with Torax Medical in the development of the LINX Reflux Management System, and Mayo Clinic licensed related technology to the company in exchange for equity. Drs. DeVault and Smith are paid consultants to Torax Medical.
Source-Newswise