‘Targeting the ‘hyperactive’ zinc transporter activity may find new ways to lower the risk of type 2 diabetes.’
Tweet it Now
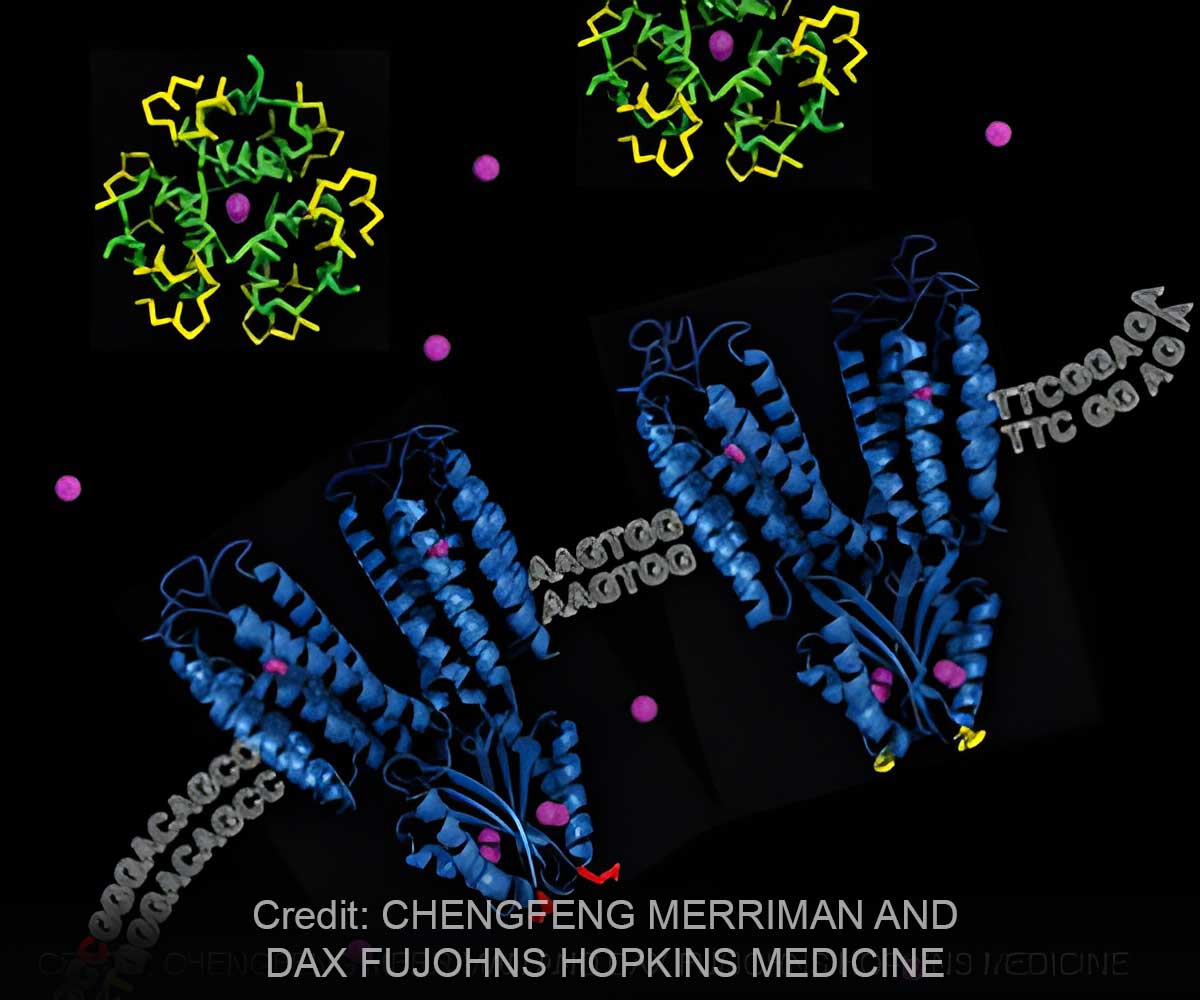
In a report on the biochemistry of the "hyperactive" genetic variant, which transports zinc ions into cells, the investigators say the finding has clarified a long-standing question about the workings of the protein the gene makes. The study will appear as a Paper of the Week in the Dec. 30 issue of the Journal of Biological Chemistry."Normally it's loss of gene function that causes disease, not enhanced function, so our finding goes against the usual assumption," says Chengfeng Merriman, Ph.D., a research specialist who was part of the Johns Hopkins study team and the report's first author. "Based on our findings, we hope that drugs can be developed that safely make the more common form of the protein behave like the less efficient form, potentially preventing many cases of type 2 diabetes."
Scientists who study type 2 diabetes generally believe the disorder is caused by a combination of many genes with relatively small individual effects, acting together with nongenetic factors, like diet, a sedentary lifestyle, age and high blood pressure.
Nearly a decade ago, separate large-scale human genome studies identified variants in a gene called SLC30A8 with a relatively big influence: People with the more common version of the gene, dubbed R, had a 12 percent greater likelihood of developing the disease than did people with the less common "W" form. Oddly enough, another study found that people with a very rare, inactive form of the gene had very low diabetes risk.
"Given that type 2 diabetes is a growing global epidemic, it has long seemed that a drug targeting the protein SLC30A8 makes, namely ZnT8 or zinc transporter 8, would potentially have a huge impact," says Dax Fu, Ph.D., associate professor of physiology at the Johns Hopkins University School of Medicine. "The problem was that studies on ZnT8 hadn't clarified whether the protein needed to be souped up or slowed down to reduce risk."
Advertisement
"In this case, breaking the 'speed limit' brings disease risk," Fu says.
Advertisement
Source-Eurekalert