‘Pirfenidone causes severe cases of drug-induced liver injury. It is strictly recommended to monitor liver function in patients receiving pirfenidone.
’
Read More..Tweet it Now
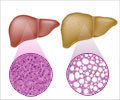
The first patient presented with acute liver failure and grade 3 hepatic encephalopathy (decline in brain function as a result of severe liver disease). The Liver function tests (liver enzymes) were elevated, the patient suffered from jaundice and had an altered mental status. Read More..
Pirfenidone was stopped in the patient. This patient had no other cause for liver failure. This patient subsequently developed organ failure and died. Post-mortem liver biopsy revealed findings of drug induced liver injury.
The second patient showed elevated liver enzymes (transaminases) after one year of pirfenidone treatment and three days after starting concomitant treatment with esomeprazole. Both the medications were discontinued, but the patient was hospitalized with jaundice, pruritis (itchy skin), nausea, vomiting and fatigue. The levels of the liver enzymes increased further. Liver biopsy was done which indicated drug induced liver injury. This patient died of sepsis, lactic acidosis and multiorgan failure.
The current alert advices to perform Liver function tests (to estimate levels of liver enzymes like AST, ALT, ALP, bilirubin) before initiating pirfenidone. These tests are to be recommended at monthly intervals for the first six months and then to be continued every three months. A strict clinical evaluation along with the lab tests must be carried out in patients showing symptoms of liver injury like fatigue, anorexia, dark urine, jaundice and right upper abdominal discomfort.
If there is a significant elevation of liver aminotransferases or symptoms of hepatic injury, dose should be adjusted or the medicine should be discontinued. The treatment and dose titration is are done according to the upper level normal (ULN) of aminotransferases (liver enzymes).
Advertisement
Monitor closely. Other medicines causing liver toxicity to be discontinued. If this is not possible, reduce dose of pirfenidone. The dose of pirfenidone can be increased to normal once the levels of liver enzymes are back in normal range
Advertisement
Permanently discontinue pirfenidone – do not reinitiate treatment.
Higher than 5 times ULN
Permanently discontinue pirfenidone – do not reinitiate treatment.
Source-Medindia