"Our study provides the first evidence that a sensory impairment, such as inner-ear dysfunction, can induce specific molecular changes in the brain that cause maladaptive behaviors traditionally considered to originate exclusively in the brain," said study leader Jean M. Hébert, Ph.D., professor in the Dominick P. Purpura Department of Neuroscience and of genetics at Einstein.
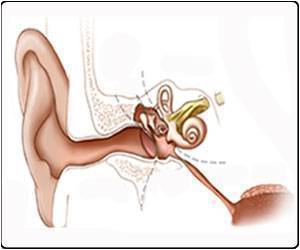
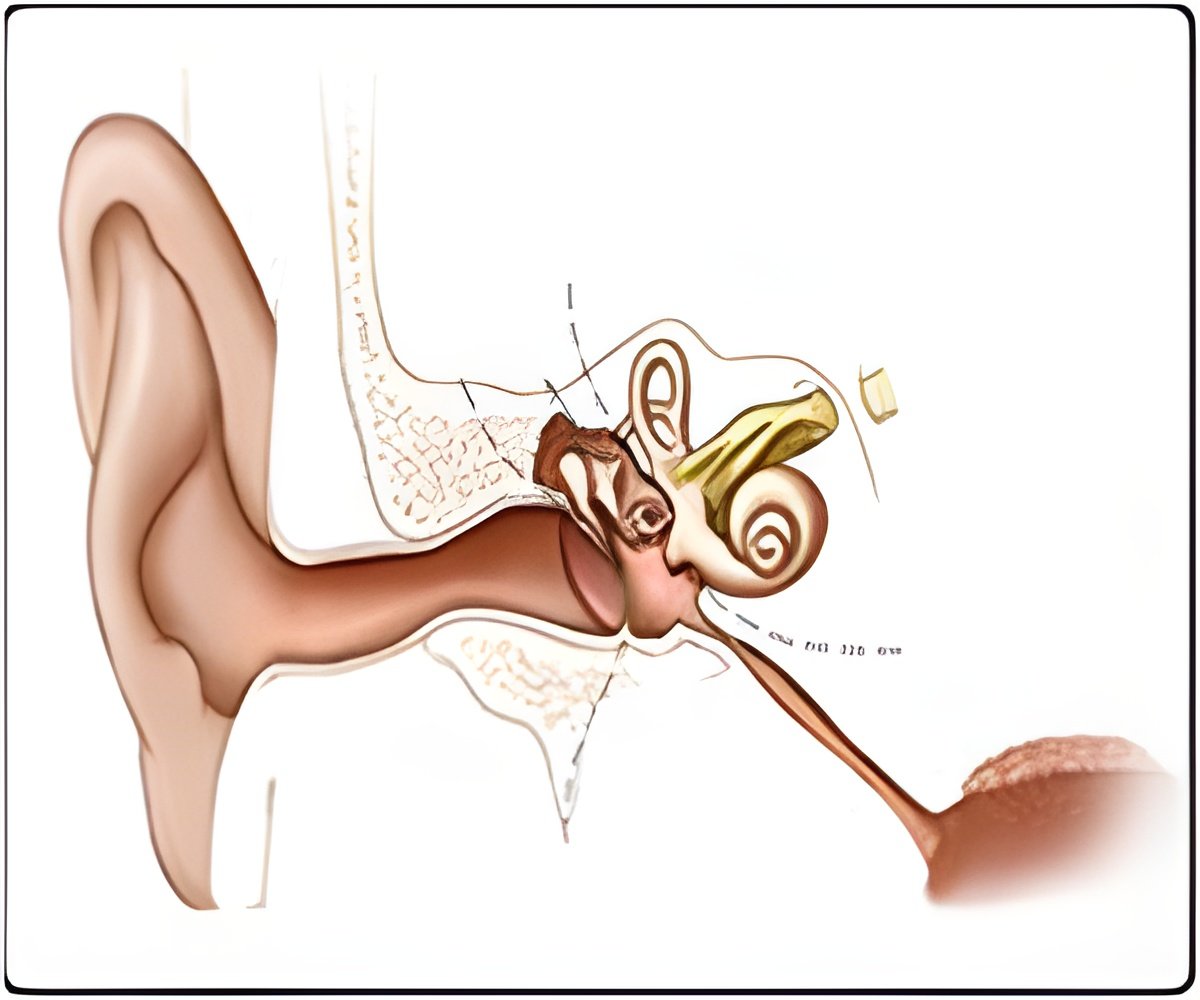
The inner ear consists of two structures, the cochlea (responsible for hearing) and the vestibular system (responsible for balance). Inner-ear disorders are typically caused by genetic defects but can also result from infection or injury.
The idea for the study arose when Michelle W. Antoine, a Ph.D. student at Einstein at the time, noticed that some mice in Dr. Hébert's laboratory were unusually active – in a state of near-continual movement, chasing their tails in a circular pattern. Further investigation revealed that the mice had severe cochlear and vestibular defects and were profoundly deaf. "We then realized that these mice provided a good opportunity to study the relationship between inner-ear dysfunction and behavior," said Dr. Hébert.
The researchers established that the animals' inner-ear problems were due to a mutation in a gene called Slc12a2, which mediates the transport of sodium, potassium, and chloride molecules in various tissues, including the inner ear and central nervous system (CNS). The gene is also found in humans.
To determine whether the gene mutation was linked to the animals' hyperactivity, the researchers took healthy mice and selectively deleted Slc12a2 from either the inner ear, various parts of the brain that control movement or the entire CNS. "To our surprise, it was only when we deleted the gene from the inner ear that we observed increased locomotor activity," said Dr. Hébert.
Advertisement
To discover whether increased pERK levels caused the abnormal increase in locomotor activity, Slc12a2-deficient mice were given injections of SL327, a pERK inhibitor. Administering SL327 restored locomotor activity to normal, without affecting activity levels in controls. The SL327 injections did not affect grooming, suggesting that increased pERK in the striatum selectively elevates locomotor activity and not general activity. According to the researchers, the findings suggest that hyperactivity in children with inner-ear disorders might be controllable with medications that directly or indirectly inhibit the pERK pathway in the striatum.
Advertisement
Source-Eurekalert