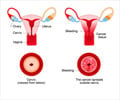
Cervical cancer screening needs to take into account a partially-vaccinated population and new technologies, according to an editorial published in the latest edition of Medical Journal of Australia.
Dr Annabelle Farnsworth, from Douglass Hanly Moir Pathology, said that although Australia has an enviable record in the control of cervical cancer, there are real benefits in incorporating new knowledge and associated technologies into screening and management of the disease.“A national, well-funded and organised program of screening using the conventional Pap Smear has significantly reduced the incidence and mortality rates of cervical cancer in Australia,” she said.
Dr Farnsworth said that there has been much discussion overseas about replacing cervical cytology tests with human papillomavirus (HPV) tests for primary screening.
“Currently, there is no justification for this as HPV testing is highly sensitive but not specific.
“HPV testing may have a greater role in the management of indeterminate abnormalities detected by cervical cytology tests.
“Both HPV tests and ThinPrep imaging [a newer type of cytology test] are more expensive than conventional cytology, but they could be cost-effective if used appropriately in conjunction with a comprehensive review of the cervical screening program,” she said.
Advertisement
“Pharmaceutical Benefits Advisory Committee commented that there would be cost savings if vaccination fully replaced cervical screening but the cervical cancer life-time risk would increase to 1.173 per cent.
Advertisement
The Medical Journal of Australia is a publication of the Australian Medical Association.
Source-MJA
SRM