‘Mutations in PUMILIO1 gene that results in neurodegeneration similar to SCA1 in animal models also has a similar affect on humans.’
Tweet it Now
These findings provide a better understanding of the genetic basis of neurological diseases and suggest that identifying protein regulators would single out new candidates for disease-causing genes. "In this study, we investigated the possibility that our findings in mice - that mutations in Ataxin1 regulator Pumilio1 cause a neurological disease similar to SCA1 - would translate to people," said first author Dr. Vincenzo Gennarino, a postdoctoral fellow in Dr. Huda Zoghbi's lab at Baylor College of Medicine during the development of this work, and currently an assistant professor of genetics and development at Columbia University Medical Center.
The amount of a single protein can affect the manifestation of a disease
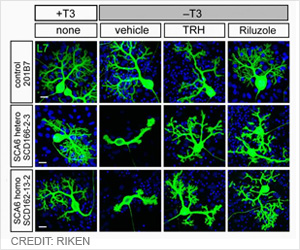
The researchers began by searching for patients with symptoms similar to those of SCA1 in large genomic databases around the world. They identified 15 patients with different mutations of PUMILIO1 gene, which they separated into two groups. In one group, the amount of PUMILIO1 protein was about half that detected in people without the condition. These patients presented with a severe form of the disease that had started early, between 5 months and 1 year of age. The children have seizures, ataxia, poor motor coordination and developmental delay. In the other group of patients, the amount of PUMILIO1 was 75 percent of that present in individuals without the condition. These patients presented with a milder form of ataxia that appeared in their 40s or 50s.
"This study, which is a nice example of the power of collaborations, taught us that mutations in the same gene can cause vastly different neurological problems based on how much they compromise the protein product," said corresponding author Dr. Huda Zoghbi, professor of molecular and human genetics and of pediatrics and neuroscience at Baylor and director of the Jan and Dan Duncan Neurological Research Institute. Zoghbi also is an investigator at the Howard Hughes Medical Institute.
Advertisement
These findings also have implications for other neurodegenerative conditions such as Alzheimer's disease, amyotrophic lateral sclerosis, or Lou Gehrig's disease, and Parkinson's disease. Identifying the regulators of proteins involved in these diseases would provide a list of candidate genes potentially linked to the condition and open new venues for novel therapeutic strategies.
Advertisement
Source-Eurekalert