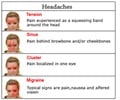
Two studies released today, offering hope for people with migraine, will be presented at the American Academy of Neurology's 66th Annual Meeting in Philadelphia, April 26, to May 3, 2014.

Both are phase II studies, meaning larger studies are needed to confirm the results.
One study involved 163 people who had migraine from five to 14 days per month. They received either a placebo or a single IV dose of a drug called ALD403 and then were followed for 24 weeks. Those who received the drug had an average of 5.6 fewer migraine days per month, a 66-percent decrease, compared to 4.6 fewer days per month for those who received a placebo, or a 52-percent decrease. Sixteen percent of those who received the drug had no migraine days at 12 weeks, while none of those who received the placebo were free from migraine at that point.
There were no differences in side effects between those receiving the drug and those receiving the placebo.
"These results may potentially represent a new era in preventive therapy for migraine," said Peter Goadsby, MD, PhD, of the UC San Francisco and a member of the American Academy of Neurology, who is an author on both studies.
"Migraine remains poorly treated, and there are few effective and well tolerated treatments approved that prevent attacks from occurring," said David Dodick, MD, of Mayo Clinic Arizona in Phoenix and a member of the American Academy of Neurology, who was also an author on both studies. "There is a huge treatment need for migraine - the third most common and seventh most disabling medical disorder in the world."
Advertisement
Those who received the drug had an average of 4.2 fewer migraine days per month at 12 weeks, or a 63-percent decrease, while those who received placebo had 3 fewer migraine days per month, or a 42-percent decrease. Those who received the drug were more likely to have side effects including pain at the injection site, upper respiratory tract infections and abdominal
pain, but overall the drug was considered to be safe and well-tolerated.
Advertisement
The ALD403 study was supported by Alder Biopharmaceuticals. The LY2951742 study was supported by Arteaus.
Learn more about migraine at www.aan.com.
The American Academy of Neurology, an association of more than 27,000 neurologists and neuroscience professionals, is dedicated to promoting the highest quality patient-centered neurologic care. A neurologist is a doctor with specialized training in diagnosing, treating and managing disorders of the brain and nervous system such as Alzheimer''s disease, stroke, migraine, multiple sclerosis, brain injury, Parkinson''s disease and epilepsy.
For more information about the American Academy of Neurology, visit www.aan.com or find us on facebook, twitter, plus.google.com and YouTube.
Editor''s Note:
Drs. Dodick and Goadsby will present their findings at 12:00 p.m. ET, on Thursday, May 1, 2014, in the Terrace Ballroom of the Pennsylvania Convention Center in Philadelphia.
Drs. Dodick and Goadsby are available for advance interviews as well. Please contact Rachel Seroka, [email protected], to schedule an advance interview.
To access Non-Emerging Science abstracts to be presented at the 2014 AAN Annual Meeting, visit www.aan.com. Emerging Science abstracts are embargoed until 12:01 a.m., ET, Friday, April 25, 2014, unless otherwise noted by the Academy''s Media and Public Relations Department.
Source-Newswise