Highlights
- Current robotic arm prosthetics rely on twitching the remnant muscles in their shoulder or arm, which are often damaged.
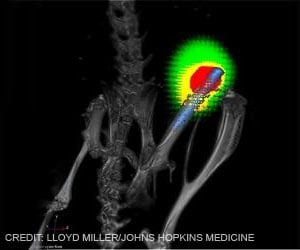
- A new sensor technology has been developed for prosthetic arms that relies on signals sent from spinal motor neurons and not on remnant muscle fiber.
- The user has to perform simple manoeuvres like grasping or pinching to control the new sensory technology based robotic prosthetics .
Using Motor Neuron Signals
Currently, robotic arm prosthetics are controlled by the user twitching the remnant muscles in their shoulder or arm, which are often damaged.
To control the sensory based prosthetic, the patient has to think like they are controlling a phantom arm and imagine some simple manoeuvres, such as pinching two fingers together.
In this technology, the user has to only perform one or two simple grasping commands.
This means that more signals can be detected by the sensors connected to the prosthetic and ultimately more commands could be programmed into the robotic prosthetic, making it more functional.
What the Researchers Did
The researchers recruited six volunteers who were either amputees from the shoulder down or just above the elbow.
To re-route parts of their Peripheral Nervous System (PNS) that were connected with hand and arm movements to healthy muscles in their body the volunteers underwent a surgical procedure. This re-routing was either directed to the pectoral muscle in the chest or the bicep in the arm, depending on the type of amputation.
The team could then clearly detect the electrical signals sent from the spinal motor neurons.
They encoded specific motor neuron signals as commands into the design of the prosthetic. They then connected a sensor patch on the muscle that had been operated on as part the re-routing procedure, which was connected to the prosthetic.
The volunteers were trained on how to control the device by thinking about specific phantom arm and hand commands by physiotherapists.
After the training they were able to move elbow joint and do radial movements like moving the wrist from side to side as well as opening and closing the hand.
The amputees were able to make a more extensive range of movements than would be possible using a classic muscle-controlled robotic prosthetic.
Finally the user was left with the basic hand and arm functions of a real arm.
Conclusion
The researchers suggest the current model is likely to hit the market in the next three years.
The next step of research will involve further refining the technology with extensive clinical trials with a much wider cross section of volunteers to validate the technology.
The study is published in the journal Nature Biomedical Engineering.
Reference
- Dario Farina et al. Man/machine interface based on the discharge timings of spinal motor neurons after targeted muscle reinnervation. Nature Biomedical Engineering ; (2017)
Source-Medindia