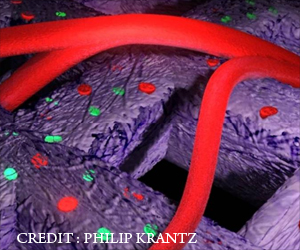
- New bioprinting technology has been developed that can create replacement organs
- This technology can be used to print intricate 3D structures that resemble human tissues
- These 3D printed tissues and organs could be used for transplantation in the future
The study was led by Dr. Jordan S. Miller, Ph.D. and Dr. Kelly Stevens, Ph.D. Dr. Miller is an Assistant Professor in the Department of Bioengineering at the George R. Brown School of Engineering, Rice University, Houston, Texas, USA. Dr. Stevens is an Assistant Professor in the Department of Bioengineering at the University of Washington, Seattle, Washington, USA.
The study was conducted in collaboration with Duke University, Durham, North Carolina, Rowan University, Glassboro, New Jersey, and Nervous System, a generative design studio in Somerville, Massachusetts, USA.
“One of the biggest roadblocks to generating functional tissue replacements has been our inability to print the complex vasculature that can supply nutrients to densely populated tissues,” said Miller. “Further, our organs actually contain independent vascular networks - like the airways and blood vessels of the lung or the bile ducts and blood vessels in the liver. These interpenetrating networks are physically and biochemically entangled, and the architecture itself is intimately related to tissue function. Ours is the first bioprinting technology that addresses the challenge of multivascularization in a direct and comprehensive way.”
In this regard, Stevens indicated that multivascularization is important because structure and function are always interrelated.“Tissue engineering has struggled with this for a generation,” Stevens said. “With this work, we can now better ask, ‘If we can print tissues that look and now even breathe more like the healthy tissues in our bodies, will they also then functionally behave more like those tissues?’ This is an important question, because how well a bioprinted tissue functions will affect how successful it will be as a therapy.”
Read More..
Why is Bioprinting So Important?
Bioprinting is a very powerful technology that can be used to print human tissues and organs. Since there is an ever-increasing demand for human organ transplantation, the power of this robust technique can be harnessed to create bioprinted organs that could, one day, reduce the reliance on natural human organs for transplantation. Moreover, transplantation of bioprinted organs will alleviate the need for lifelong treatment with immunosuppressants, which is currently the norm. In fact, the researchers are optimistic that the application of bioprinting in medicine could become a reality within the next two decades.How were the Bioprinted Organs Fabricated?
The research team bioprinted the intricate components of the human liver and lungs using a new open-source bioprinting technology called Stereolithography Apparatus for Tissue Engineering (SLATE). This technology utilizes additive manufacturing for making soft hydrogels that are constructed layer-by-layer.Each layer is printed using a liquid pre-hydrogel solution that solidifies upon exposure to the blue light generated by a digital light projector. Each of the layers, which are roughly 10-50 microns in thickness, is sequentially added on top of each other to create the 3D structure of the tissue or organ.
This technique allows the production of soft, water-based, biocompatible gels that can be moulded into intricate internal structures of organs within a matter of minutes. In this regard, the bioengineers from Nervous System were instrumental in designing the internal architecture of the tissues and organs.
What did Bioprinted Organ Testing Reveal?
Tests were carried out on bioprinted lung alveoli and liver tissue, which are indicated below:- Lung Alveoli: The tests revealed that the lung-mimicking air sacs (alveoli) were stable and capable of carrying air without bursting. Since these alveolar sacs were pulsatile, they allowed the rhythmic flow of air that closely resembled human breathing. The tests also revealed that the red blood cells were capable of taking up oxygen during their passage through the network of blood vessels that surrounded the alveolar sacs. Importantly, the movement of oxygen closely resembled the gas exchange that occurs in the actual lung alveoli.
- Liver Tissue: In case of liver tissue, tests were carried out in vivo in a mouse model of chronic liver disease. The 3D printed liver tissues, which had separate compartments for blood vessels and liver cells, were loaded with primary hepatocytes and implanted into mice. The tests revealed that the liver cells survived the implantation process
Future Prospects of Bioprinting Technology
The newly developed bioprinting technology has tremendous prospects in the medical field. The technology can be applied for developing structures such as bicuspid valves present in the heart, which allows blood to flow only in one direction. Other types of valves present within the leg veins, and associated lymphatic vessels can also be printed using this technology. Thus, bioprinting these multivascular and intravascular structures introduce extensive design freedoms for engineering living tissues, which makes it possible to build a great variety of intricate structures present in the human body.The new bioprinting technology is currently being commercialized through a start-up company called Volumetric. This Houston-based company designs and manufactures bioprinters and bioinks.
Concluding Remarks
Since an open-source model was used for developing the 3D bioprinting technology, all data are freely available in the public domain for use by anyone wanting to build their own stereolithography printing apparatus and hydrogels.In this regard, Miller said: “Making the hydrogel design files available will allow others to explore our efforts here, even if they utilize some future 3D printing technology that doesn’t exist today.” He added: “We are only at the beginning of our exploration of the architectures found in the human body and still have so much more to learn.”
Funding Source
The study was funded by the National Institutes of Health (NIH), the National Science Foundation, the Robert J. Kleberg, Jr. and Helen C. Kleberg Foundation, the John H. Tietze Foundation, and the Gulf Coast Consortia.Reference:
- Multivascular Networks and Functional Intravascular Topologies within Biocompatible Hydrogels - (https://science.sciencemag.org/content/364/6439/458)
Source-Medindia