Highlights:
- Lower levels of protein in the eye increases intraocular pressure, leading to glaucoma, finds a new research
- Injecting an antibody ABTAA (Ang2-binding and Tie2-activating antibody) in the eye reduces intraocular pressure and reverse the progression of glaucoma
- The findings could be a promising therapeutic option for glaucoma
The Development and Progression of Glaucoma
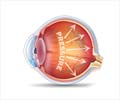
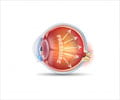
Abnormally high pressure inside the eye is one of the most important risk factors for glaucoma. Aqueous humor is a transparent watery fluid which is constantly produced and drained out from the eye. It transports nutrients and oxygen to the cells and inflates the eye giving it a roughly spherical shape. But, when the fluid cannot flow out of the eye chambers freely, an intraocular pressure increases, which damages the optic nerve, leading to vision loss.The mechanism of elevated resistance to aqueous humor outflow remains unclear. The current treatments for glaucoma tackle the production and outflow of aqueous humor, but their outcomes are still poor.
Schlemm’s canal is a component of the eye that plays a key role in draining out the aqueous humor. The canal collects the aqueous humor and mediates its transfer from the eye chambers. The endothelial cells on the walls of the Schlemm’s canal transfer the fluid from the inner to the outer side in packages called ’vacuoles.’ The shape and number of the vacuoles reflect the outflow performance. Thus, several giant vacuoles are expected in the normal outflow process.
Imbalance in Schlemm’s Canal Increases Risk of Glaucoma
The research team explained how imbalances in Schlemm’s canal significantly increase the risk of glaucoma. Angiopoietin-Tie2 is an important regulator for canal functionality. Angiopoietins Ang1 and Ang2, are proteins important for the growth of new blood vessels and angiopoietin-Tie2 is a receptor that binds them.Angiopoietin-Tie2 system plays a critical role in Schlemm’s canal formation. Tie2 mutations or the absence of angiopoietin can result in congenital glaucoma. However, this also plays a role in the development of glaucoma during adulthood.
The research team conducted a study on mice to study the development of glaucoma. They found that adult mice deficient in Tie2 showed an elevated intraocular pressure, retinal neuronal damage and partial visual impairment.
They also found a decreased number of giant vacuoles inside Schlemm’s canal endothelial cells, which indicate a poor aqueous humor outflow.
The research team also investigated how this process occurs in older mice because aging is also a major risk factor for glaucoma. They found that older mice experience reduced levels of giant vacuoles, Tie2, Ang1, and Ang2. Other proteins that are connected with the angiopoietin-Tie2 pathway, like Prox1 were also reduced in older mice.
New Treatment for Glaucoma
The research team tested if the activation of Tie2 could reverse the condition. They injected an antibody ABTAA (Ang2-binding and Tie2-activating antibody) in one eye of mice, while the other eye of the same mice was the negative control.After one week of observation, the research team found that the eye treated ABTAA had increased levels of Tie2, Prox1 and the number and diameter of giant vacuoles in the Schlemm’s canals also increased when compared to the control eye.
The intraocular pressure in the eye also decreased after ABTAA was injected into the eyes of mice suffering from POAG with regressed Schlemm’s canals.
The findings indicate that the antibody ABTAA could be a therapeutic option for treating glaucoma.
"Slow development of glaucoma treatments is partly due to the poor understanding of the underlying pathogenesis. We hope that identifying the critical role of the angiopoietin-Tie2 system in adult Schlemm’s canals will bring a significant boost in the development of therapeutics," said KOH Gou Young, the corresponding author of the study. "
Glaucoma
Glaucoma is the second leading cause of irreversible blindness, affecting about 3.5% of the world population aged 40 to 80.The two major types of glaucoma are
- Primary open-angle glaucoma
- Angle-closure glaucoma
- Early treatment could help prevent glaucoma
- Anyone could be at risk for glaucoma
- There are no warning symptoms
- People of Asian descent are at a higher risk for angle closure glaucoma
- People of African or European origins are at greater risk for primary open angle glaucoma
- The prevalence of glaucoma in Southern India is 2.6%, however, 90% of these cases have never been diagnosed early
- Angle-closure glaucoma has reached epidemic proportions in China and some parts of Asia
Reference:
- Glaucoma is second leading cause of blindness globally - (http://www.who.int/bulletin/volumes/82/11/feature1104/en/index1.html)
- Jaeryung Kim, Dae-Young Park, Hosung Bae, Do Young Park, Dongkyu Kim, Choong-kun Lee, Sukhyun Song, Tae-Young Chung, Dong Hui Lim, Yoshiaki Kubota, Young-Kwon Hong, Yulong He, Hellmut G. Augustin, Guillermo Oliver,and Gou Young Koh1. Impaired Angiopoietin/Tie2 Signaling Compromises Schlemm’s Canal Integrity and Induces Glaucoma. Journal of Clinical Investigation (2017). DOI:https://doi.org/10.1172/JCI94668DS1
Source-Medindia