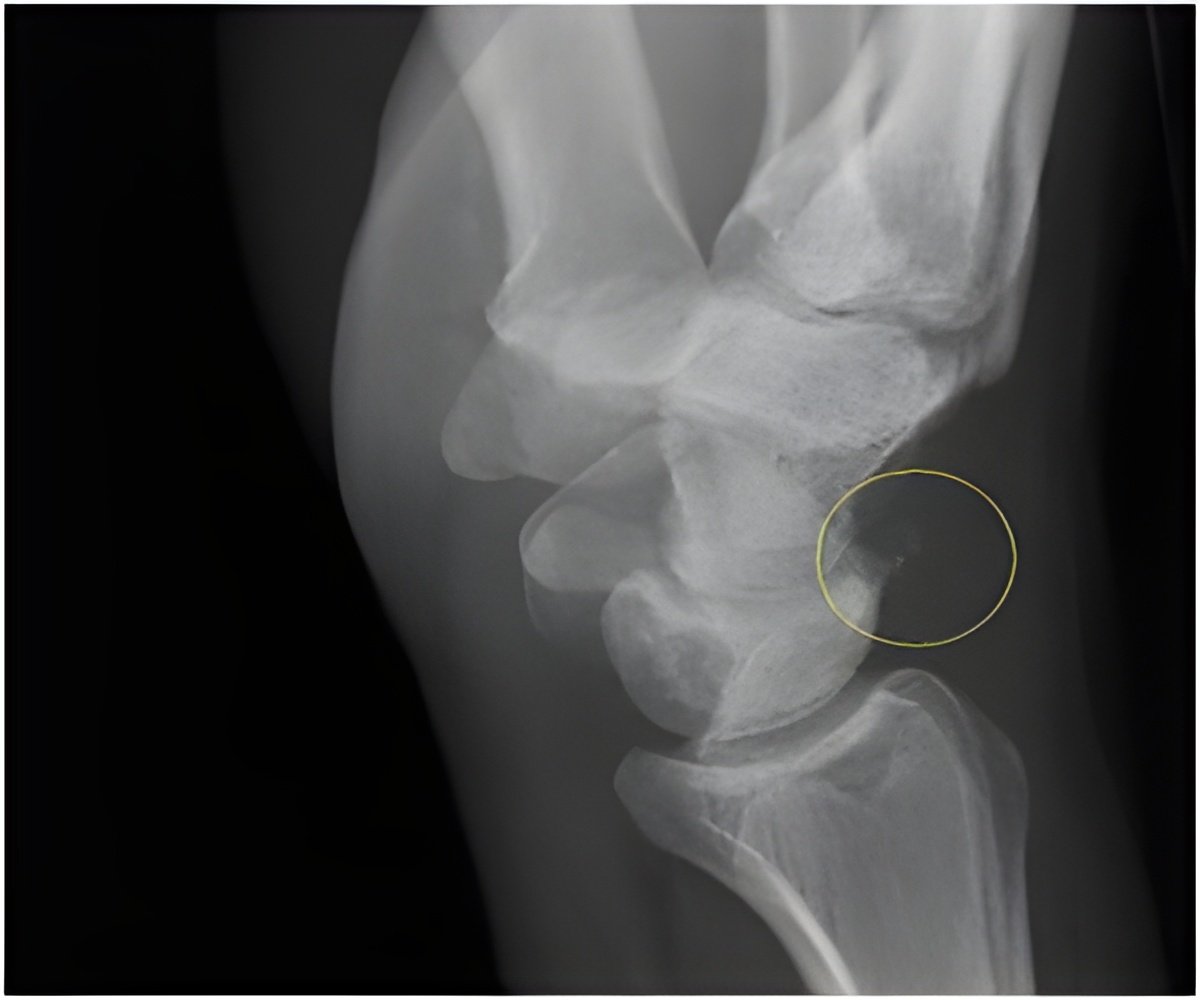
‘Receiving low intensity pulsed ultrasound (LIPUS) to speed up bone healing after fracture has little or no impact on pain or recovery time.’
Tweet it Now
Each device costs between US$1300 and $5000 and data suggest it is
commonly used in clinical practice. But some studies have shown that the
potential benefits of LIPUS on bone healing are highly uncertain.New evidence suggests that receiving low intensity pulsed ultrasound (LIPUS) to speed up bone healing after fracture has little or no impact on pain or recovery time, say a panel of international experts in The BMJ today.
They say LIPUS does not represent an efficient use of health resources and recommend that it should be stopped.
Their advice is part of The BMJ's 'Rapid Recommendations' initiative - to produce rapid and trustworthy guidance based on new evidence to help doctors make better decisions with their patients. Both the new evidence and the guidance are published by The BMJ today.
So The BMJ's guideline panel - made up of bone surgeons, physiotherapists, clinicians and patients with experience of fractures - carried out a detailed analysis of the latest evidence.
Advertisement
As such, they unanimously recommend against LIPUS for patients with any bone fractures or osteotomy (the surgical cutting of a bone to allow realignment).
Advertisement
It is unlikely that new trials will alter the evidence, they add. And they suggest that future research "should focus on other interventions that have a greater probability to speed up healing."
Source-Eurekalert