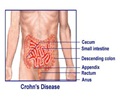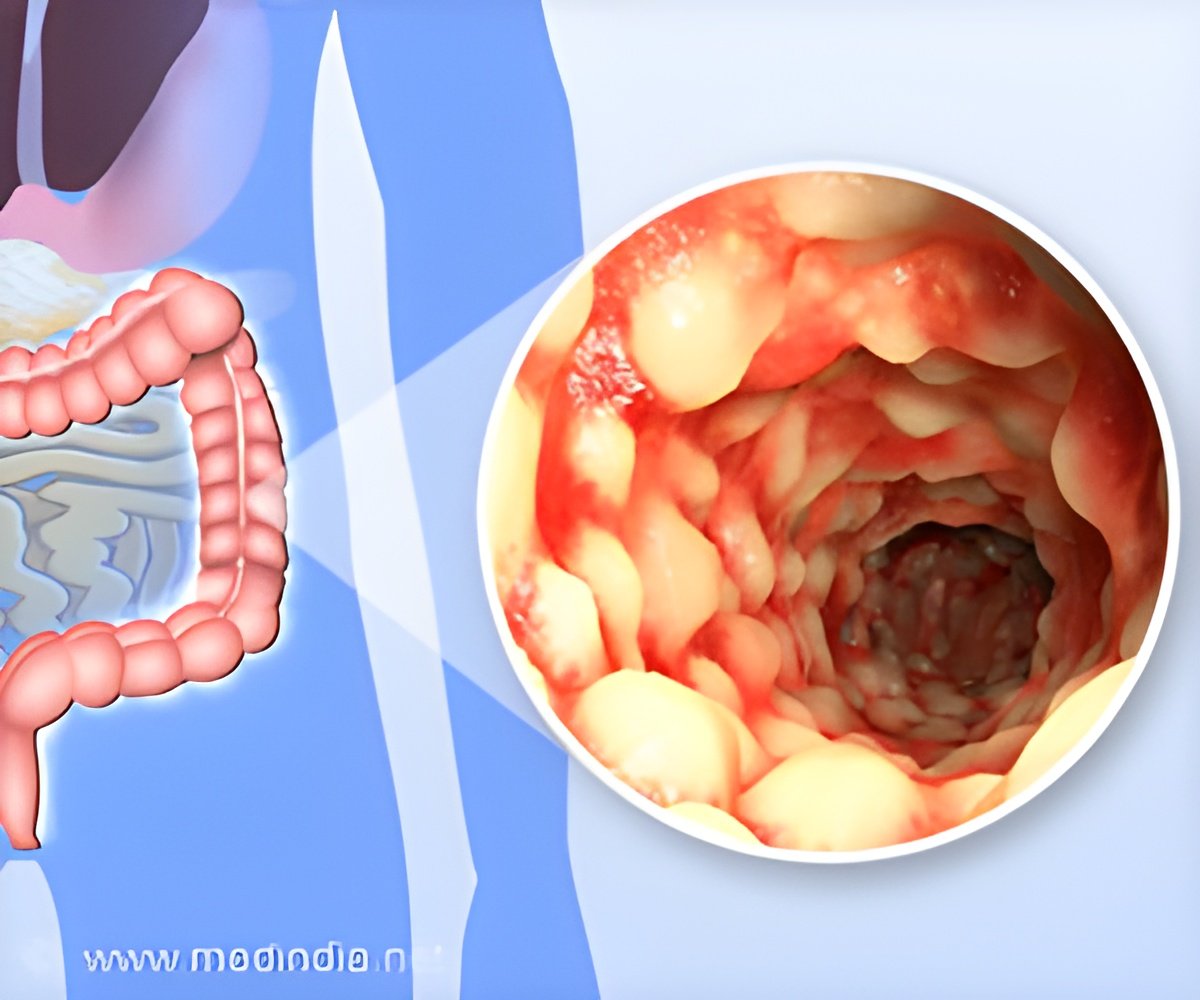
The trials are being conducted at multiple clinical sites in North America and Europe by its subsidiary company Glenmark Pharmaceuticals SA.
Glenmark Pharmaceuticals Chief Scientific Officer Michael Buschle revealed that the phase II trials will be expanding the sample base to 100-300 people. “Despite treatment advances in recent years, UC remains a debilitating condition for many individuals, and represents an area of substantial unmet medical need”, he said.
Source-Medindia