- Retinoblastoma is the most form of eye cancer.
- Early diagnosis, when tumors are small, maximizes survival.
- Genetic counseling and screening is the new basis of screening for retinoblastoma.
Systematic screening of children at elevated risk because of family history of retinoblastoma has dual purposes:
- To provide a method for detecting disease at the earliest possible stage.
- To focus care on the children at highest risk, while decreasing unnecessary evaluations for children at low or no risk above that of the general population.
Screening children early
Children who are considered to be at risk of developing eye cancer should receive genetic counseling and testing as soon as possible to clarify risk for the disease. The goal is to ensure retinoblastoma is detected at the earliest possible stage so ophthalmologists can save the lives and vision of more children.
Retinoblastoma in children
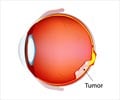
Retinoblastoma is a cancer that starts in the retina, the very back of the eye. It can also spread to other parts of the body, including the brain and bones. There are approximately 350 new cases diagnosed each year in the United States.
The disease primarily affects young children. It can be either hereditary or non-hereditary. Children with hereditary retinoblastoma often develop retinal tumors in both eyes within the first years of life. Early diagnosis, when tumors are small, improves the child’s chance of survival and their chance of keeping their vision and their eyes.
Development of these guidelines began when ophthalmologist Alison Skalet, M.D., Ph.D., of the Casey Eye Institute in Portland, Ore., searched for an optimal screening strategy for her own patients and found little published guidance.
The guidelines address a knowledge gap among ophthalmologists and other health care professionals in the U.S. regarding risk for inherited retinoblastoma and best practices for screening examinations. It is anticipated that they will also influence care in other countries. Therefore, the guidelines were written to provide a general framework for care that can be modified based on local resources, and provider and parental preferences. The recommendations acknowledge pediatric anesthesia and genetic testing may be limited in many developing countries, preventing strict adherence. So, the guidelines offer direction in cases when these resources are unavailable.
References:
- Alison H. Skalet et al. Screening Children at Risk for Retinoblastoma, Ophthalmologyhttp://www.aaojournal.org/article/S0161-6420(17)31784-0/fulltext
Source-Eurekalert