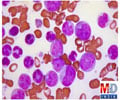
The mutation was first observed in a family treated at Memorial Sloan-Kettering of which several family members of different generations had been diagnosed with childhood acute lymphoblastic leukemia (ALL). A second, non-related, leukemia-prone family cared for at a different hospital was later found to have the same mutation. A series of experiments were conducted confirming that the observed mutation compromised the normal function of the gene, which may increase the risk of developing ALL.
The inherited genetic mutation is located in a gene called PAX5, which is known to play a role in the development of some B cell cancers, including ALL. PAX5, a transcription factor or "master gene," regulates the activity of several other genes and is essential for maintaining the identity and function of B cells. In all study participants, one of the two copies of the PAX5 gene was missing, leaving only the mutated version. The research continues as the researchers believe additional genetic factors played a role in the development of ALL in these patients.
ALL is the most common form of cancer in children, with 3,000 children and young adults being diagnosed each year in the United States.
Dr. Offit hopes that ongoing research will also determine what percentage of childhood ALL patients have the PAX5 mutation. Current estimates suggest that it is rare. Additionally, the newly discovered gene mutation may someday help scientists determine how to target transcription factors to treat other non-inherited forms of leukemia where the PAX5 mutation is present.
"With a better understanding of the genetic elements that induce cancer susceptibility, or drive cancer to grow, we can more precisely target therapy as well as potentially prevent cancer from occurring in the first place," added Dr. Offit.
Advertisement
Source-Newswise