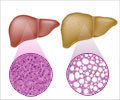
"The liver is a particularly hard-to-control bleed site during surgery, as blood loss is often higher and hemostasis may be difficult to achieve," said Krishna Athota, M.D, trauma and critical care surgeon at the University of Cincinnati College of Medicine.
Clinical studies demonstrate that EVARREST is greater than 94% effective compared to current standard of care in controlling bleeding across challenging patient types and surgical situations.
"This expanded indication for EVARREST reinforces this innovation’s potential to make problematic bleeding situations routine, and could result in a paradigm shift in the treatment of bleeding during surgery," he added.
Source-Medindia