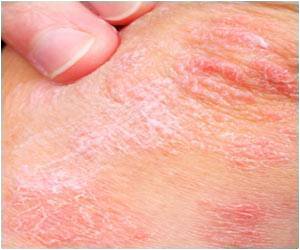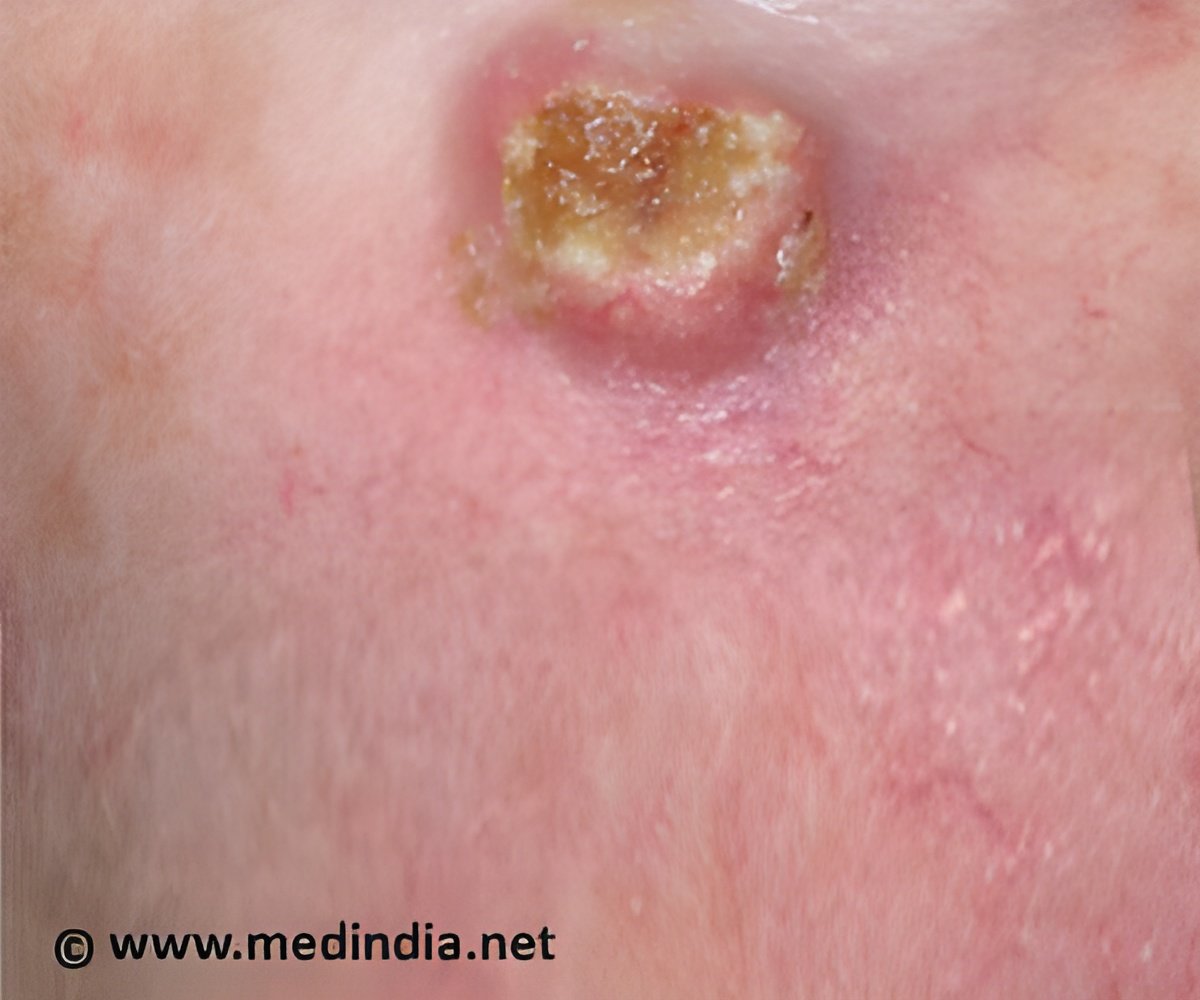
‘National Youth Risk Behavior Survey, 2013 reports that 1.6 million minors indoor tan each year which increase their risk of skin cancer and other skin damages.
’
Tweet it Now
UV radiation exposure in children and teenagers puts them at a greater risk of skin and eye damage later in life. Also, indoor tanning increases the risk for melanoma, a deadly form of skin cancer.FDA has issued two proposed rules regarding indoor tanning beds,
Proposal 1: Both Children and adults must abide these rules.
- restrict use of indoor tan beds to individuals 18 and older.
- Adults users must sign a risk acknowledgement certification before their first tanning session and every six months after that. Proposal 2: FDA has issued these rules for the tan bed manufacturers.
- To make warnings bigger on the device and fix a Panic button for emergency shut off.
- Limit the amount of light to the light allowed through protective eye wear
- Improving labeling on replacement bulb and prohibiting dangerous device modifications
“The FDA understands that some adults may decide to continue to use sunlamp products. These proposed rules are meant to help adults make their decisions based on truthful information and to ensure manufacturers and tanning facilities take additional steps to improve the safety of these devices,” said Ostroff.
Advertisement