The results came from the PANORAMA 2 clinical trial of panobinostat combined with bortezomib (Velcade) and dexamethasone in relapsed multiple myeloma patients whose disease became resistant to bortezomib.
Panobinostat is a histone deacetylase (HDAC) inhibitor, a type of drug that blocks key processes involved in gene expression and protein degradation. Panobinostat clogs up a protein disposal mechanism in myeloma cells so that harmful byproducts accumulate and eventually cause programmed cell death.
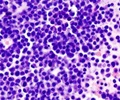
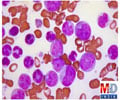
Abnormal cells in multiple myeloma accumulate in the blood, bones and other organs, causing them to malfunction. The effectiveness of a multiple myeloma drug can be evaluated by the magnitude of its effect on the amount of disease present in the body, typically assessed by measuring serum monoclonal protein.
The PANORAMA 2 trial included 53 patients with relapsed multiple myeloma who had undergone multiple rounds of prior treatment and, in more than half, also stem cell transplant. The researchers reported on 44 patients receiving the panobinostat-bortezomib-dexamethasone combination.
Results showed that in the first phase of the treatment, 9 of the patients had at least a partial response of their disease, and 2 of the 9 saw their myeloma almost disappear, a so-called near complete response. Another 7 patients experienced minimal response, which is also associated with clinical benefit.
Eight patients have advanced to a second phase of treatment, and 6 of them are still on treatment as much as 8 months later, says Dr Richardson.
Side effects included fatigue, diarrhea, low platelet counts, nausea and anemia, but Richardson says that these problems have proven manageable with supportive care and dose-reduction.
Advertisement