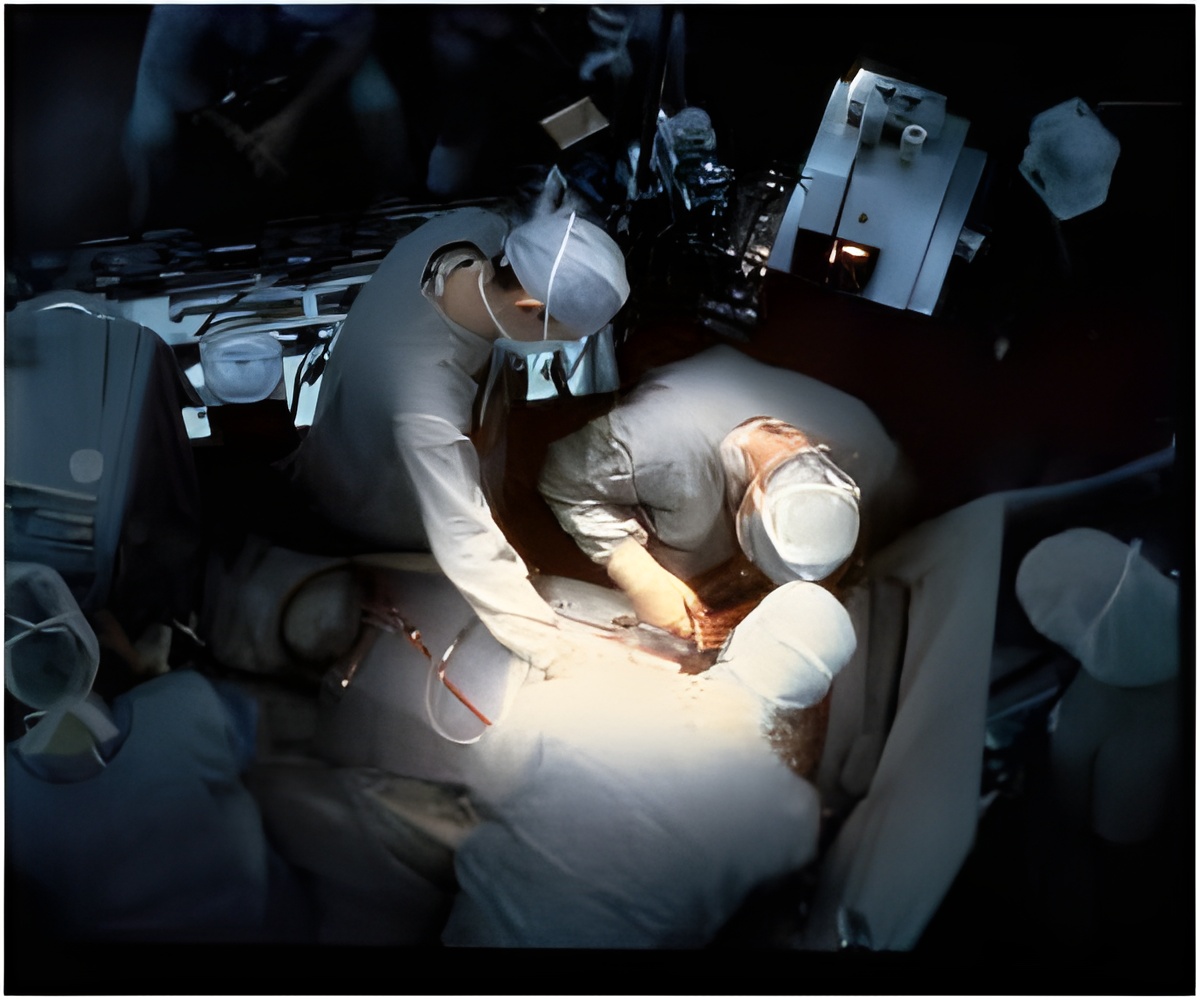
NeuroVive Pharmaceutical and Hospices Civils de Lyon (HCL) today announced the enrollment and treatment of the first patient in the European multicenter trial of myocardial infarction (the CIRCUS study). NeuroVive's advanced CicloMulsion(TM) cremophor-free IV cyclosporine formulation is used in this study of 1,000 patients undergoing percutaneous coronary intervention (PCI) for acute myocardial infarction to examine cyclosporine's ability to protect cardiac tissue. The double-blind, placebo-controlled, investigator-initiated study is being led by trial sponsor Professor Michel Ovize, MD, PhD, of HCL. NeuroVive is supporting the study by providing active drug, placebo and drug logistics. The study will enroll patients at 40 centers in France and additional European sites.
Professor Michel Ovize comments:"Patients with myocardial infarction often undergo emergency percutaneous coronary intervention in which a catheter passed through the major blood vessels restores blood flow to blocked coronary arteries. Even after blood flow is restored, damage to the heart muscle continues to progress through what is known as reperfusion injury. Small-size proof-of-concept studies have shown that reperfusion injury may account for as much as 30-40% of total infarction-related myocardial damage and that timely therapeutic interventions can prevent this damage (Staat et al. Circulation 2005; Thibault et al. Circulation 2008). From a clinical point of view, it is critical to develop a drug to protect cardiac tissue during PCI and hopefully improve clinical outcome."
NeuroVive CSO Associate Professor Eskil Elmer comments:"Professor Ovize showed previously in a study published in the New England Journal of Medicine (NEJM, 2008 Jul 31; 359 (5): 473-81) that cyclosporine reduces reperfusion injury by approximately 40% in patients with myocardial infarction. If, as we expect, findings are reproduced in this larger cohort of patients with significant improvement in patient outcomes, the impressive capability of cyclosporine as a cardiac tissue protective agent will be confirmed."
NeuroVive´s CEO Mikael Bronnegard comments: "If efficacy is proven, NeuroVive´s CicloMulsion(TM), a safe formulation of cyclosporine, should become an important new treatment for heart patients and reduce morbidity and mortality after myocardial infarction. In addition, it will deliver a major reduction in the social and economic burden of heart disease and target a significant and growing global market of patients with cardiovascular disease."
Source-Eurekalert