Researchers have found that an experimental drug called raxibacumab has been found to be effective in treating anthrax poisoning.
"The results published today showed that a single dose of raxibacumab was highly effective as a treatment for inhalation anthrax in both rabbits and monkeys," said Dr Sally D. Bolmer, R.A.C, lead author and Senior Vice President, Development and Regulatory Affairs, HGS."Raxibacumab acted quickly to provide a significant survival benefit to animals showing clinical signs of disease caused by exposure to a dose of aerosolized anthrax spores that was approximately 200 times the median lethal dose.
"We also note that the safety profile shown in healthy human volunteers provides support for use of raxibacumab in the clinical setting of immediately life-threatening inhalation anthrax disease," she added.
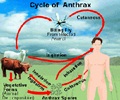
The drug works by targeting anthrax toxins after they are released by the bacteria into the blood and tissues.
In an inhalation anthrax attack, people may not know they are infected with anthrax until the toxins already are circulating in their blood, and it may be too late for antibiotics alone to be effective.
"We are very proud that the importance of these data and the rigor and high quality of our scientists' work have led to publication in The New England Journal of Medicine," said Dr David C. Stump, Executive Vice President, Research and Development, HGS. Based on these results, we believe raxibacumab has the potential to be a significant step forward in the treatment of inhalation anthrax," he added.
Advertisement
Source-ANI
TAN