‘Brain-derived neurotrophic factor (Bdnf) deficiency-induced diabetes can be reversed by duodenum-jejunum gastric bypass surgery.’
Tweet it Now
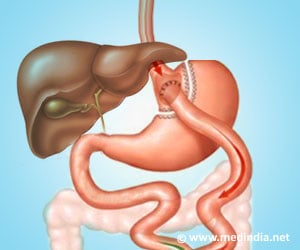
"Our research showed that duodenum-jejunum gastric bypass (DJB) surgery may be applied to cure diabetes of both genetic (mutation) and environmental (diet-induced) origin," explained lead investigator Xiang Gao, PhD, of State Key Laboratory of Pharmaceutical Biotechnology and MOE Key Laboratory of Model Animal for Disease Study, Model Animal Research Center, Nanjing Biomedical Research Institute and the Collaborative Innovation Center of Genetics and Development, Nanjing University.
"We found that DJB surgery induced gut microbiota alterations, which may be the key reason for diabetes remission after bariatric surgery. Our data indicate that suppressed inflammation is the result, not the cause, of diabetes reversal in these genetically modified mice." The research was performed in the T2DM mouse model that mimics key symptoms including insulin resistance, high blood levels of lipids, metabolic inflammation, and obesity. These mice harbor genetic mutation in brain-derived neurotrophic factor (Bdnf) leading to Bdnf deficiency. Bdnf is a member of the neurotrophic family of growth factors and is a key regulator of both brain function and metabolic balance.
"Our findings suggest that Bdnf deficiency-induced diabetes can be reversed by DJB surgery in mice, which has potential for the treatment of diabetes in humans," stated Dr. Gao. He and his team found that bypass surgery reversed the metabolic abnormalities indicative of diabetes without changing Bdnf expression directly. Glucose tolerance and insulin sensitivity were greatly improved and there was less fat accumulation in liver and white adipose tissue. Insulin sensitivity reached normal levels within two weeks following surgery and lasted for at least eight weeks. Six weeks after bypass surgery, oral glucose tolerance in the treated mice was significantly lower than in the diabetic mice that had undergone a sham operation and was similar to levels observed in untreated controls.
Examination of the composition of bacteria and other microorganisms in the gut of mutated mice before and after bypass surgery and in the control group, showed a decrease in pathogenic bacteria and an increase in beneficial microflora that coincided with the onset of better glycemic control. "More mechanistic studies of gut microbiota alterations after bypass surgery are needed to explain how different families of microbiota may regulate nutrient metabolism in the host," noted Dr. Gao.
Inflammation, especially in white fat tissue and liver, is thought to play an important role in obesity and T2DM. Eight weeks after bypass surgery, significant reductions in inflammatory indicators occurred in the liver and fat tissue, although the post-surgical anti-inflammatory effects occurred after insulin sensitivity improved. "These results indicate that the alleviation of inflammation was not the direct cause of the improvement in insulin sensitivity that resulted from bypass surgery," commented Dr. Gao.
Advertisement
Source-Eurekalert