When the brain develops in the uterus, new brain cells are born deep within the brain, near the center. These newborn brain cells then crawl out of the so-called "niche" where they were born and migrate outward to the edges of the brain. This outermost layer of the brain is known as the cerebral cortex and is the seat of all higher-level thinking and cognition.
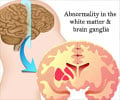
In girls with a mutation on their X chromosome, the outward migration of brain cells unfortunately fails. Instead of making it all the way to the edges of the brain, some of the brain cells pile up on top of one another and form a secondary or "double-cortex." The activity of these abnormally placed brain cells gives rise to seizures and also, in some cases, mental retardation.
Drs. Bechstedt and Brouhard were able to purify the product of the mutated gene, a protein known as doublecortin, and to watch the protein in action under a microscope. This protein helps brain cells to build a scaffold inside themselves, much like the scaffolds at construction sites, built of "poles" called microtubules; these form a "skeleton" for the brain cells, known as the cytoskeleton. Brain cells require this internal skeleton to crawl and to migrate, much as humans need their skeletons to walk and run.
The McGill researchers discovered that, in order for doublecortin proteins to help build this scaffold, many doublecortin proteins must work together as a team. They found that disease-causing mutations cause a breakdown in this teamwork. This loss of teamwork is sufficient to prevent the brain cells from constructing a proper "skeleton."
This discovery has implications for treatments for a range of conditions, from other forms of epilepsy to spinal cord injuries. In each case, therapies are increasingly directed at triggering brain cells to extend their skeletons -- for example when re-growing a nerve ending past the site of a wound in the spinal cord. Understanding how brain cells construct their skeletons will open avenues for doctors to target the brain cell skeleton to extend and re-grow when needed.
Advertisement