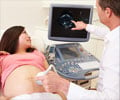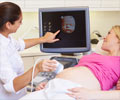
These two products are the first in Carestream’s new family of ultrasound systems, which earned praise for its all-touch control panel and advanced imaging capabilities when demonstrated to attendees of the 2014 Radiological Society of North America conference and the 2015 American Institute of Ultrasound in Medicine meeting.
“These new ultrasound systems build upon our proven expertise in digital X-ray systems. We started with a clean slate and worked with customers to develop a new generation of ultrasound systems that enhance ease of use and productivity, while incorporating integrated GPU processing power that delivers fast response times and extremely high image quality,” said Diana L. Nole, President, Digital Medical Solutions, Carestream.
Both Touch Ultrasound systems deliver these capabilities:
- A sealed panel allows for easy cleaning to help limit the spread of pathogens
- High-level computing power provides both speed and excellent image quality
- Easy maneuverability and a small, lightweight footprint make the imaging process faster and easier
- “Swipe and go” badge log-on saves time and promotes secure access
- “Smart connect” transducer technology enables easy one-touch selection of the desired transducer
- Rapid cold boot time of 18 seconds can further help enhance productivity, with no need for standby mode or battery backup
Source-Medindia