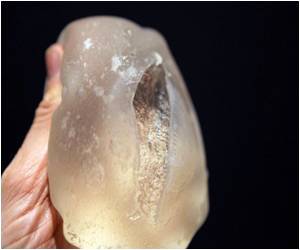
Surgeons in Britain had warned against the use of the PIP implants in 2006 and 2007, but it took regulators a further four years to ban them.
An investigation by the government showed that the rupture rate could be seven times higher than British regulators previously believed.
The evidence was supplied privately to the Department of Health (DoH) on Friday by Transform, Britain's biggest cosmetic surgery chain.
The findings prompted the government to question the official figures supplied to the regulators by private cosmetic surgery companies.
The Health Secretary, Andrew Lansley, was reportedly 'very concerned' and 'unhappy' that firms had not given the regulator full information about the failure rate.
Advertisement
At least 3,000 NHS patients - most recovering from breast cancer - were given the faulty implants as part of reconstructive surgery.
Advertisement
Transform's rupture rate of seven per cent - if replicated across the industry - is seven times higher than the one per cent official figure the British regulators was sticking to last week in its attempt to reassure tens of thousands of women.
"One of our surgeons is helping the DoH. He has taken our data with him and we believe - and it needs rechecking - that the rupture rate is actually higher at about seven per cent," the Telegraph quoted a source at Transform as saying.
The source said the figures had been supplied to the Medicines and Healthcare Products Regulatory Agency (MHRA) since 2006 and questioned why the regulator had not picked up on them sooner.
In December, the MHRA declared there was 'no evidence of any disproportionate rupture rates other than in France'.
In France, the official rupture rate is five per cent, and women who have the implants are being offered free removal.
Other cosmetic surgery providers in the UK have now been ordered to comb through their data.
Source-ANI