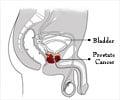
Active surveillance or watchful waiting might be a suitable treatment option for patients with prostate cancer that has a low risk of progression, a new study has said.
Treatment of localized prostate cancer is controversial because, for some, this disease will not progress during their lifetime, and treatment may incur serious and long-lasting side effects.An increasingly popular option is active surveillance, or deferring treatment until evidence of disease progression.
To investigate outcomes of patients treated with active surveillance, Par Stattin, of the Department of Surgical and Perioperative Science at Umea University, and colleagues conducted an observational study of 6849 patients in the National Prostate Cancer Register of Sweden with localized prostate cancer who were 70 years old or younger.
The patients had low or intermediate risk of prostate cancer progression and were treated with active surveillance or watchful waiting; or radical prostatectomy or radiation therapy from 1997 through December 2002.
In this cohort, 2021 patients received surveillance, 3399 received radical prostatectomy, and 1429 received radiation therapy. After a median follow-up of 8.2 years, there were 413 deaths in the surveillance group; 286 in the radical prostatectomy group, and 1429 patients in the radiation therapy group.
The researchers found a much higher percentage of death from competing causes in the surveillance group (19.2 percent, compared with 6.8 percent in the prostatectomy group and 10.9 percent in the radiation therapy group), suggesting that patients with a shorter life expectancy were more often selected for surveillance than surgery or radiation therapy.
Advertisement
However, the difference in absolute risk between the groups was modest, at 1.2 percent, after 10 years of follow-up.
Advertisement
"With a 10-year prostate cancer-specific mortality of less than 3 percent for patients with low-risk prostate cancer on surveillance, this strategy appears to be suitable for many of these men," they said.
The study has been published online June 18 in The Journal of the National Cancer Institute.
Source-ANI


![Prostate Specific Antigen [PSA] Prostate Specific Antigen [PSA]](https://www.medindia.net/images/common/patientinfo/120_100/prostate-specific-antigen.jpg)