‘Drug-resistant infection is responsible for killing more than 700,000 people worldwide every year. India is no exception to the increasing cases of antibiotic resistance and drug resistance bacteria.’
Tweet it Now
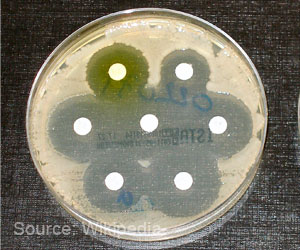
Expressing concern over the growing resistance against antibiotics in the country, the Indian Medical Association (IMA) has come out with a draft policy on use of antibiotics in human healthcare aiming at reining in antimicrobial resistance (AMR). As per the policy, which applies to all the IMA members in the country, antibiotics should not be prescribed for fever with rash, cough or cold, suspected or confirmed dengue, malaria, chikungunya, viral hepatitis or any viral syndrome, unless clinically warranted. Any antibiotic prescribed should be put in a box, in patient prescriptions, so that patient can identify the antibiotic in his/her prescription. All prescriptions should be accompanied with a rider stating ‘no refill without doctor’s prescription’ (could be printed on the prescription pad as footer).
No newer antibiotic molecules have been are invented in last 30 years. The antibiotics that are currently available are only modifications of the existing molecules have been tried out. This is one of the reasons for antibiotic resistance and the multidrug-resistant microorganisms are a real threat to healthcare in India.
Total number of antibiotic tablets/capsules to be taken for the prescribed duration shall be mentioned in the prescription and not just the dose administration schedule. Appropriate antibiotics should be prescribed at the earliest to manage suspected sepsis, meningitis, pneumonia or positive cases of tuberculosis.
Every medical establishment can draw its own antibiotic policy (IV to oral antibiotic switch, antibiotic preference based on local antibiogram, infection prevention and control, reuse of medical devices and safe syringe practices). MDR TB and XDR TB are required to be notified to health authorities and surveillance teams (IDSP). Doctors need to ensure root cause analysis for any outbreak of MDR infection in hospital/healthcare facility. All food products must be labeled with “Antibiotic status”. Antibiotic waste disposal policy to be developed to prevent contamination of the environment; preventing discharge of untreated waste into soil and rivers, it added.
Advertisement
Source-Medindia