‘High-fat, high-sugar diet that leads to obesity, along with normal aging factor, may contribute to the development of Alzheimer's disease. Alzheimer's disease is a progressive brain disorder that leads to loss of brain functions.’
Tweet it Now
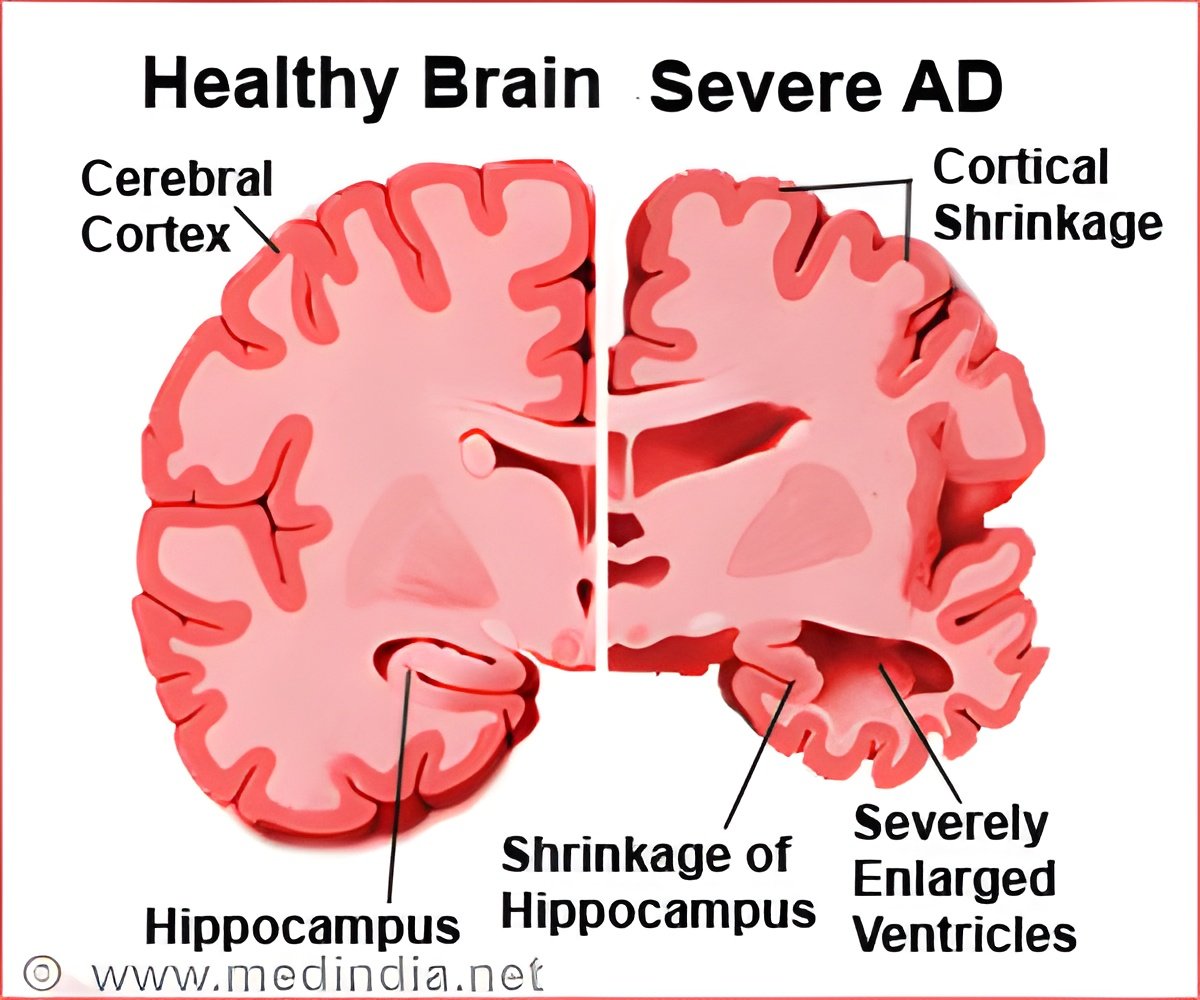
Alzheimer's disease, the most common form of dementia, is a progressive brain disorder that leads to loss of cognitive skills and memory and causes significant changes in behavior. Aging is a significant risk factor for Alzheimer's. Previous studies suggest that diet-related obesity is also associated with development of the disease. Researchers from Brock University in Ontario, Canada, looked at the effects of an obesity-inducing diet on insulin signaling (the process that tells the body how to use sugar), and markers of inflammation and cellular stress. These factors have been found to be involved in the progression of Alzheimer's disease during the aging process in mice.
One group of mice received a high-fat, high-sugar diet ("HFS"), while the control group ate a normal diet. The researchers measured the animals' inflammation and stress levels in the hippocampus and the prefrontal cortex in the brain after 13 weeks on the assigned diets. They compared the brains of aged mice to those of a younger set of baseline mice. The hippocampus is near the center of the brain and is responsible for long-term memory. The prefrontal cortex, at the front of the brain, oversees complex cognitive, emotional and behavioral function.
Compared to the control group, the HFS group had significantly higher markers of inflammation, insulin resistance (altered insulin signaling) and cellular stress in areas of the hippocampus thought to be involved in the progression of Alzheimer's disease. The prefrontal cortex region of the HFS group showed more signs of insulin resistance, but inflammation and cellular stress markers did not change. The "region specific differences between the prefrontal cortex and hippocampus in response to aging with a HFS diet [indicates] that the disease pathology is not uniform throughout the brain," the researchers wrote.
The control group's inflammation levels were also increased after the trial when compared to the baseline readings. These results support the theory that aging alone plays a role in the progression of Alzheimer's disease, and obesity increases the effects of aging on brain function.
Advertisement
Source-Eurekalert