New information about the mechanism that leads to the destruction of brain cells following a stroke, as well as a possible way to mitigate that damage has been revealed by researchers at the Brain Research Centre.
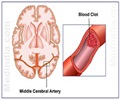
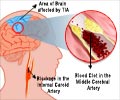
After a stroke, many brain cells continue to die even after blood flow has been restored.Researchers have long known that this is due to a complicated cascade of cellular messages that lead to the "self-destruction" and death of brain cells.
In the new study, it was discovered that, in animal models, the over-activation of NMDA receptors-special receptors on the surface of brain cells-activates another protein, called SREBP-1, which subsequently causes cell death.
SREBP-1 is found naturally in cells throughout the body and is involved with cholesterol and other fat production.
NMDA receptors control the movement of calcium in and out of brain cells, which is necessary for normal brain function.
But, after a stroke, levels of glutamate-the most abundant chemical messenger in the brain-rise rapidly in cells, leading to over-activation of NMDA receptors, an excess of calcium entering cells, and the onset of cell death.
Advertisement
After a stroke, over-activation of NMDA receptors leads to a rapid degradation of Insig-1, which increases the level of active form of SREBP-1.
Advertisement
While the detailed mechanisms by which activation of SREBP-1 leads to brain cell death remain to be established, the researchers discovered a way to inhibit SREBP-1 and thereby significantly reduce cell death.
"We developed a drug that can stabilize Insig-1, which in turn inhibits the activity of SREBP-1," said Dr. Max Cynader, co-author of the study.
The researchers also found that the drug works post-stroke in animal models.
"When we administered it post-stroke, there was less brain cell damage 30 days later than compared to controls," says Dr. Wang. "This is important because previous studies focused on blocking the NMDA receptors in order to prevent cell death, but this approach didn't work because it affected normal cell function and had a relatively short therapeutic window. The drug we studied works downstream of NMDA receptors and appears to have less detrimental side effects with a much improved therapeutic window."
The study has been published online in Nature Medicine.
Source-ANI
RAS