Two separate studies have shown that human mesenchymal stem cell (hMSC) and bone marrow stromal cell (BMSCs) transplantation, respectively, may be beneficial for stroke victims.
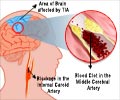
Both studies tracked the migration of chemically “tagged” transplanted stem cells to determine the degree to which the transplanted cells reached damaged areas of the brain, and became therapeutically active.One of the studies was conducted by Korean researchers, wherein hMSCs were transplanted into animal stroke models with cerebral artery occlusion and tracked by magnetic resonance imaging (MRI) at two days, one week, two weeks, six weeks and ten weeks after transplant.
“Cells started showing indications of migration as early as one or two weeks following transplantation. At 10 weeks, the majority of the cells were detected in the core of the infarcted area,” said lead author Jihwan, Song, DPhil, of the Pochon CHA University College of Medicine.
The researchers concluded that there is a strong tendency for transplanted hMSCs to migrate toward the infarcted area regardless of injection site, but that the degree of migration was likely based on differences in each animal’s ischemic condition.
“We speculate that the extensive migratory nature of stem cells and their utilization will provide an important tool for developing novel stroke therapies,” said Song.
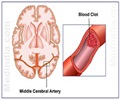
In the second study, jointly conducted by Canadian and Chinese researchers, BMSCs—connective tissue cells—were injected into animals 24 hours following middle cerebral artery occlusion.
Advertisement
“We evaluated vascular density in the ischemic region in all animals seven days after cell transplantation. The animals exhibited significant reductions in scar size and cell death and improvements in neurological function when compared to controls that received no BMSCs,” said study lead author Dr. Ren-Ke Li.
Advertisement
Though the potential mechanisms for the recovery are unclear, the researchers believe that one of the possibilities is that the brain microenvironment early on following a stroke may mimic brain development.
Subsequent elevated levels of growth factors might enhance homing of BMSCs to the injured area and induce cell proliferation, they say.
“Our results support the potential therapeutic use of BMSCs after a stroke,” Li concluded.
“Both studies lend important support to a growing body of laboratory evidence that bone marrow is a remarkable adult stem cell source for transplant therapy following stroke,” says Cell Transplantation associate editor Cesar V. Borlongan, Ph.D. of the Medical College of Georgia.
“The non-invasive MRI visualization of pre-labeled BMSCs could become a routine clinical marker for transplanted cells as well as for safety and efficacy,” Borlongan added.
Both studies have been published in the journal Cell Transplantation.
Source-ANI
LIN/K