Rutgers University researchers have made a discovery that may help introduce significant improvement in cochlear implants.
Cochlear implants today operate with varying degrees of success in different patients. While some facilitate the hearing of sounds like the rush of traffic or the crash of thunder, others provide even better hearing response such as detecting voice and understanding speech while still being unable to appreciate music.Robin Davis, a professor in the Department of Cell Biology and Neuroscience at the university, says that his team’s work may provide for a dramatic improvement in such implants.
He says that the study’s findings are important for engineers and surgeons in designing new cochlear implants.
“The significance of our work lies in the fact that we can change an element in a very peripheral part of the sensory system that can have an impact all the way into the brain,” he said.
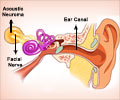
Cochlear implants, also known as “bionic ears”, are surgically inserted into the snail-shell shaped structure within the inner ear called the cochlea. Hair cells line the cochlea and convert acoustic signals into electrical signals that nerves then carry to the brain.
Davis says that sounds can be amplified with a hearing aid where some hair cells exist. But where the hair cells are missing or damaged, a condition generally associated with severe hearing impairment, an implant may be used to replace their function, he adds.
Advertisement
“Our studies have revealed that spiral ganglion auditory neurons possess a rich complexity that is only now beginning to be understood,” said Davis.
Advertisement
Begun over six years ago, Davis’ research is now producing insights into precisely how these multidimensional proteins operate in the cochlea.
A report on their most recent findings, published in The Journal of Neuroscience, says that while neurotrophins have historically been prized for the survival value they impart to nerve cells, the researchers have found that in the cochlea they do a great deal more.
Their presence in relative proportions transforms the spiral ganglion neurons into either fast-firing transmitters to carry high-pitched sound messages to the brain, or slow-firing carriers for the transmission of lower pitched signals.
The neurotrophins accomplish this at the molecular level by tightly regulating a newly defined and complex series of signalling proteins, says the report.
According to Davis, one end of the cochlea is home to the slower-firing neurons characterized by a preponderance of NT-3, while the other cochlear end is rich in BDNF, making those neurons faster-firing.
The researcher says that both neurotrophins are present in gradients throughout the range, but at any specific locale their amounts vary relative to each other – lots of BDNF and a little NT-3 in the high frequency transmitters, for example, and the reverse as you move toward the other end.
In one possible remedial approach, Davis described how the neurotrophins could potentially be pumped into a newly designed cochlear implant and released through graduated ports along its length.
Source-ANI
SRM/P