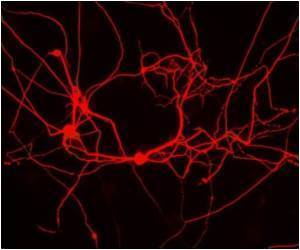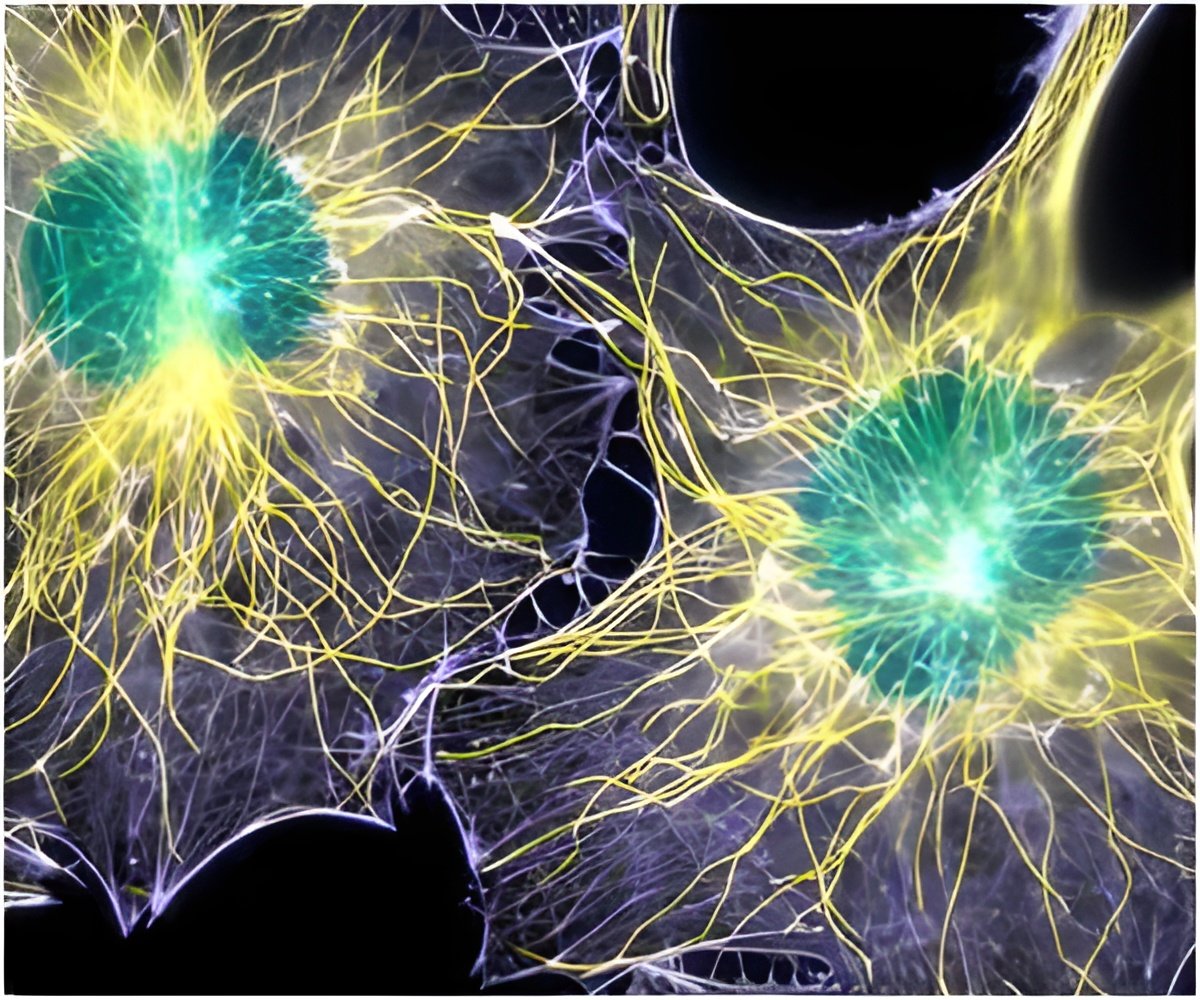
An interdisciplinary team of Queen's University researchers led by Dr. Lauren Flynn, a professor in the Departments of Chemical Engineering and Anatomy and Cell Biology, has been working with stem cells extracted from samples of human fat and is developing new methods in the lab to develop these cells into mature tissue substitutes.
While stem cells extracted from fat cannot be grown into as many different types of cells as embryonic stem cells, they do have a number of advantages.
"The advantages include less ethical controversy, abundant cell availability from discarded tissues from elective surgeries like breast reductions and tummy tucks, and a much reduced possibility for immune rejection when re-implanting cells extracted from a person's own fat," explained Dr. Juares Bianco, a postdoctoral fellow in the Department of Chemical Engineering and the Human Mobility Research Centre (HMRC) who is working in the Flynn lab group.
Sarah Fleming, a Master's candidate in the group, is also working to establish a new method for growing the fat stem cells in the lab using a system that mimics the natural tissue environment found within the body.
This work is based on Flynn's development of a technique for washing away all traces of cells from a sample of body fat, leaving behind a three-dimensional tissue scaffold that she calls "decellularized adipose tissue", or "DAT" for short.
Advertisement
Flynn's preliminary studies have shown that when the stem cells are grown on the DAT scaffold, they naturally begin to mature into fat cells, suggesting that the engineered growth environment influences the type of cell that the basic stem cells will turn into during the tissue regeneration process.
Advertisement