A method that can manage the amount of fat that cells store for use as a back-up energy source has been discovered by scientists. It can eventually lead to therapies for liver disease and diabetes.
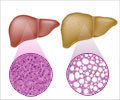
Researchers at Albert Einstein College of Medicine of Yeshiva University have also found that the process, if disrupted, can enable cellular fat to accumulate, a key factor in age-related metabolic diseases such as obesity and type 2 diabetes.They reckon that the discovery of this previously unknown fat-fighting pathway may lead to novel drugs for the treatment of metabolic syndrome, characterized by obesity, blood lipid disorders, and insulin resistance, and for a common liver disease known as "fatty liver" or steatohepatitis.
Usually, all cells store lipids, a type of fat, in the form of small droplets that can be broken down for energy when needed.
But in cases of excessive food intake, or in certain diseases like diabetes or obesity, these lipid droplets become so large that they interfere with normal cell function.
"In this study, we found that the amount of fat stored in these intracellular lipid droplets is controlled through autophagy, a process until now thought to help primarily in digesting and recycling damaged cellular structures," Nature magazine quoted Dr. Mark Czaja, professor of medicine at Einstein, as saying.
Autophagy, also known as the process of "self-eating", is carried out by lysosomes, which function as the cell's recycling center.
Advertisement
"When food is scarce, autophagy becomes a main source of energy for the cells and this process of digesting lipid droplets is accelerated. If autophagy slows down, as occurs in aging, the lipid droplets stored in cells keep growing and eventually become so big that they can no longer be degraded," said a co-author of the study.
Advertisement
The researchers noted that therapies aimed at helping autophagy operate more efficiently might prevent disease by keeping fat droplets under control.
Source-ANI
ARU/L