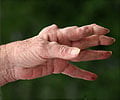
Anakinra, a new breed of arthritis drugs, has some modest beneficial effect for rheumatoid arthritis patients, but may also have potential risks for serious infections, according to a new study.
The study also warned against its use with other biologic medications.Rheumatoid arthritis is a chronic inflammatory disease affecting as many as one in 100 people worldwide. It affects the joints, making them stiff and painful.
Belonging to drugs licensed in the past decade called 'biologics', anakinra is the first to target the immune protein IL-1, thought to be partly responsible for inflammation in arthritis patients. It is given by daily injection.
After conducting a Cochrane Systematic Review of five recent anakinra trials, involving 2,876 patients, the researchers found that the drug reduced pain and stiffness in patients, and helped to improve joint function, when compared to placebo.
While around one-fourth of patients experienced improvement in their symptoms as a result of taking anakinra, it was found that the improvements were notably less than those seen with other biologics.
Besides, there were more injection site reactions with the use of anakinra and also the rate of serious infections with anakinra was touching statistical significance when compared to placebo.
Advertisement
One study included in the review investigated the combination of anakinra with another biologic medication, etanercept, and was found to have no benefits in arthritis outcomes.
Advertisement
"On the basis of these results, we recommend that doctors avoid combining biologic medications with anakinra when treating patients with rheumatoid arthritis,' said Mertens.
Source-ANI
PRI/SK