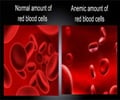
Researchers have identified the specific biological mechanisms believed to lead to a rare and incurable blood disease known as Diamond Blackfan anemia (DBA). Scientists say with further investigation, their discoveries could result in drastic changes to current thinking about treatment for this disease and may lead to promising new drug therapies.
George Thomas, PhD, Stefano Fumagalli, PhD, and collaborators report their findings online ahead of print in the journal Nature Cell Biology on Sunday, March 15, 2009. The research will also appear in the April print issue of the journal and is being presented at the 10th annual International Diamond Blackfan Anemia Consensus Conference in New York, which concludes Monday, March 16.DBA is a rare blood disorder characterized by the bone marrow's failure to produce red blood cells. This failure is due to an intrinsic defect that makes the red blood cells prone to cell death before they mature. Red blood cells travel through the bloodstream to deliver oxygen to the body's tissues, which is critical to the health and proper function of all tissues.
According to the Centers for Disease Control and Prevention, approximately 25-35 new cases of DBA are diagnosed each year, with the majority of patients being identified before age 1. The most common treatments include blood transfusions and corticosteroids. The disease is characterized by extreme anemia—with a propensity to develop into leukemia—and often has no cure.
Using a preclinical laboratory model, Thomas' team was able to explain how cell death occurs in DBA and identified a specific step in the biological chain of events leading to disease onset where targeted medical intervention may effectively slow—or even stop—red blood cell death.
DBA has recently been attributed to a ribosomal protein defect that the UC team hypothesizes leads to abnormal activation of p53, causing premature death of red blood cells. P53 is a protein that normally functions to trigger "cell suicide" in response to severe cellular damage, therefore protecting the body from overgrowth of defective cells.
Previous research has attributed p53 activation to the passive diffusion of ribosomal protein L11 from the nucleolus, the part of the nucleus where ribosomes are produced to the nucleoplasm.
Advertisement
"Previous studies suggested L11 was passively coming out of the nucleolus when ribosome production was disrupted. Our study actually showed that the nucleolus stayed intact as ribosomes were still being produced, suggesting selective upregulation of L11," explains Thomas, the John and Gladys Strauss endowed professor of cancer biology at UC and scientific director at UC's Genome Research Institute. "If we can target the L11 interaction, we might be able to spare other stress pathways that mediate potential benefits of p53 induction."
Advertisement
"By understanding the chain of biological events leading to this abnormal cell death and targeting the specific molecular checkpoint that controls cell death, we may be able to develop new drugs that would interrupt or stop the process and allow the body to recover, rebuilding healthy bone marrow," adds Thomas.
Source-Eurekalert
SRM