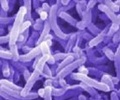
The National Institute of Immunology (NII), New Delhi is in a process of developing a vaccine to safeguard children from diarrhea caused from rotavirus.
Some international pharmaceutical companies like GSK have developed vaccine against rotavirus but their efficiency under Indian conditions is not known."We have been working on developing another vaccine which is based on an Indian strain of virus which is rotavirus 116 E. This strain of virus was isolated from All Indian Institute of Medical Science (AIIMS) from children who were born in the hospital," said Dr. Sudhanshu Vrati, senior scientist, NII.
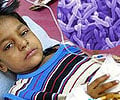
The medical authorities are now gearing up for phase III clinical trial of this vaccine that will confirm its efficiency.
"The vaccine was tested on 90 children. We tested two doses of vaccine. At the lower dose of the vaccine about 60 percent of the children zero converted whereas in the higher dose of vaccine close to 90 percent of children zero converted. Whether this zero conversion will result to reduced diarrhea that is yet to be seen," said Dr. Sudhanshu Vrati, senior scientist, National Institute of Immunology, New Delhi.
"Now we are gearing up for phase III clinical trial of this vaccine which will tell the efficacy of the vaccine," he added.
Rotaviral infections are the leading cause of diarrhea- associated mortality in developing countries.
Advertisement
Rotaviral diarrhea contributes 40 percent of the total dehydrating diarrhea disease in younger children causing 150000 deaths per year.
Advertisement
The department of Biotechnology is supporting a rotaviral diarrhea vaccine development project since 1990-1991 under Indo-US vaccine action programme.
Source-ANI
SRM